In this article I will cover the less well known, albeit highly significant, NO generating process, the nitrate-nitrite-NO pathway. This new NO producing pathway holds a lot of promise and supplements that target it will probably replace the current arginine based NO boosters in the near future[6, 7].
The nitrate-nitrite-NO pathway is especially interesting in that it not only has performance enhancing effects in healthy folks – as well as in people with risk factors – but also offers cardiovascular protection, regardless of baseline health status [7-10].
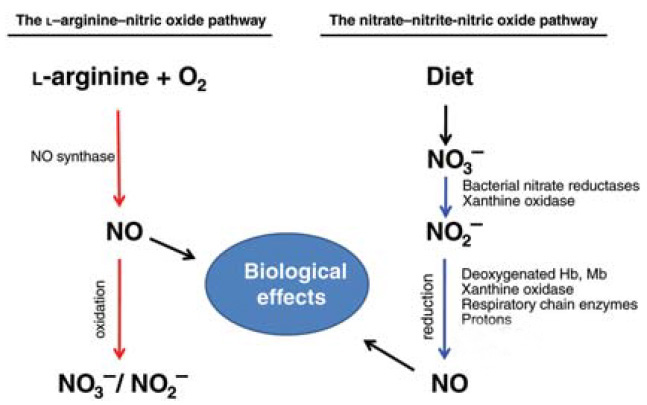
Nitric oxide production from the arginine-eNOS-NO pathway is dependent on the activity of the eNOS enzyme, which itself is controlled by other factors. In contrast to the arginine-eNOS-NO pathway, which forms the basis of current NO boosting supplements, nitrate-nitrite-NO pathway produces NO by a mechanism that does not involve the eNOS enzyme.
As indicated in figure 1, NO production via the nitrate-nitrite-NO pathway starts by conversion of nitrate to nitrite, which is carried out by bacteria in the oral cavity. Once nitrite is formed, several pathways exist (deoxygenated hemoglobin/myoglobin, xanthine oxidase, respiratory chain enzymes etc) which convert nitrite to NO [1, 2].
Figure 1: NO production via the nitrate-nitrite-NO pathway.
Many of the biochemical reactions that drive the nitrate-nitrite-NO pathway are greatly accelerated under hypoxic and acidic conditions, such as those occurring during high intensity exercise [3-5]. This is an important point because hypoxic conditions reduce the activity of the eNOS enzyme [11], which catalyzes the production of NO from L-aginine. The importance of the nitrate-nitrite-NO pathway has been underscored by the finding that nitrite supplementation restores NO supply and is cardio-protective in mice lacking the arginine-eNOS-NO pathway [12].
Thus, the nitrate-nitrite-NO pathway is complementing the better known arginine-eNOS-NO pathway for generation of NO when the arginine-eNOS-NO pathway isn’t running smoothly. And a growing number of studies are shown that the nitrate-nitrite-NO pathway appears to be far more effective in healthy people looking for a performance enhancing edge, than the arginine-eNOS-NO pathway.
Several studies have demonstrated that nitrate supplementation reduces the oxygen cost for a given exercise intensity [13-18]. This has practical implications because exercise economy affects tolerance to high-intensity exercise [19]. Three recent studies confirm that nitrate supplementation increases tolerance to high-intensity exercise by enhancing muscle contractile efficiency and reducing metabolic perturbations in muscle [13, 16, 17]. Mechanistically, a central target for the effects of nitrate and its metabolites seems to be the mitochondrion [20].
In one study, healthy men were given 500 ml of a nitrate-rich beetroot juice providing 316 mg of nitrate, or placebo, 6 consecutive days. On the last 3 days they subjects completed low-intensity and high-intensity “step” exercise tests. On days 4-6, the nitrate-rich beetroot juice supplement resulted in a significant increase in blood nitrite levels that was more than double that seen with placebo. During low-intensity exercise, the nitrate-rich beetroot juice supplement attenuated the reduction in muscle phosphocreatine by 35% and reduced the exercise associated oxygen cost with 25%. During high-intensity exercise, time to exhaustion was 12 min 14 sec in the nitrate supplemented group, compared to 9 min 46 sec in placebo, which corresponds to a 25% increased performance. The total ATP turnover rate was significantly less for both low-intensity and high-intensity exercise in the nitrate supplemented group, compared to placebo. It was concluded that the reduced oxygen cost of exercise following nitrate-rich beetroot juice supplementation is due to a reduced ATP cost of muscle force production [16], and that the reduced muscle metabolic perturbation (as reflected in the extent of depletion of muscle phosphocreatine) allowed high-intensity exercise to be tolerated for a greater period of time [16].
Most studies also show that nitrate supplementation, when taken in amounts that sufficiently elevate blood nitrite levels, boosts performance in high-intensity exercise time trials [21-28]. More specifically, a study that investigated the effects of a single dose dietary nitrate supplementation on power output and performance during 2.5 and 10 mile cycling time trials [23]. Club-level competitive male cyclists were assigned to consume 17 oz of beetroot juice (providing 384 mg nitrate) or 17 oz of nitrate-depleted beetroot juice as placebo, 2.5 h before the cycling trials. The results showed that the nitrate supplementation significantly increased power output during both the 2.5 and 10 mile cycling time trials, and improved 2.5 mile performance by 2.8% and 10 mile performance by 2.7%. The performance increase was attributed to an improved cycling economy, as demonstrated by a higher power output for the same oxygen consumption [23].
Another study tested whether 6 days of nitrate supplementation would improve competitive time-trial performance in trained cyclists, as it does after a single dose. Male cyclists ingested 140 ml/day of concentrated beetroot juice (496 mg nitrate per day) or a placebo (nitrate-depleted beetroot juice) for 6 d. After supplementation on day 6, subjects performed 60 min of submaximal cycling (2×30 min at 45% and 65% Wmax, respectively), followed by a 6.2 mile time trial. It was found that the nitrate supplementation lowered sub-maximal oxygen consumption, and increased time-trial performance with 12 seconds (1.2%) and power output by 2 % [22].
Intake of 200 mg baked beetroot (providing 500 mg nitrate) has been shown in healthy fit men and women to increase running speed by 5% during the last 1.1 miles (1.8 km) of a 3.1 mile (5 km) treadmill run, and also reduce ratings of perceived exertion during that same exercise segment [27]. This finding supports the mechanistic theory that the nitrate-nitrite-NO pathway kicks in when that arginine-eNOS-NO pathway flakes out.
As of this writing, there are yet no long-term studies on the potential effects of nitrate supplementation in conjunction with resistance exercise training. But since the hypoxia and acidity stimulates the activity of the nitrate-nitrite-NO pathway, and knowing that nitrate supplementation improves muscle contractile efficiency, there are good reasons to believe that it may boost anaerobic muscle performance and enhance training outcomes related to muscle growth and strength.
It is especially exciting when performance enhancing supplements also confer health benefits. Nitrate, which is found in most vegetables and is particularly abundant in leafy greens and beetroot, has emerged as being one of the compounds that contribute to the cardiovascular and metabolic health benefits associated with high vegetable consumption [20, 29-38].
Nitrite, the conversion product of nitrate, is more abundant in meant (see below). While most studies on nitrite appear to show that its effects occur via generation of NO, there are also indications that nitrite itself may act as a signaling molecule and regulator of gene expression [39], and be an endocrine mediator of NO-based cellular signaling [40, 41]. Nitrite itself can also perform many actions that were previously attributed to NO [42].
The most notable and widely relevant cardiovascular protection seen with nitrate and nitrite intake is its well documented salutary impact on blood pressure and vascular function [2, 37, 43-48]. Elevated blood pressure is an established risk factor for coronary artery disease, stroke, kidney disease, all-cause mortality, and shortened life expectancy [49, 50]. Furthermore, the relation between blood pressure and health consequences is progressive throughout the non-hypertensive range [51]. Given these considerations, and the high prevalence of both hypertension and pre-hypertension (30% and 66% respectively, pre-hypertension defined as 120-139/80-89) [52, 53], even a small decrease in blood pressure may have a significant health effect. It is notable that two thirds of the American population has pre-hypertension, and that over a third of those are undiagnosed [52]. This is an important issue since hypertension causes premature aging of endothelial function and thereby accelerates development of age-related cardiovascular disease [54].
Thus, dietary modification and/or supplementation to increase nitrate and nitrite intake in order to ramp up the nitrate-nitrite-NO pathway hold tremendous benefit for the vast majority of people, of all ages (more on that below). In addition, dietary nitrite and nitrate have been shown to reduce inflammation, restore endothelial function, lower C-reactive protein levels, and protect from heart attack, stroke and type-2 diabetes [20, 55, 56]. Nitrate intake, in a dose that corresponds to a large intake of vegetables, has also been shown to reverse features of metabolic syndrome [38]. Long term nitrate ingestion may even reduce abdominal (visceral) fat and lower blood fats (triglycerides) [38].
Our diet is the main contributor to the body’s pool of nitrate and nitrate, with vegetables accounting for 60-85% of daily nitrate intake [30, 36]. Meats (both processed and unprocessed), the second food source, contain more nitrite than vegetables. Processed meat contains a little more nitrite and nitrate than un-processed meat, but the difference is much less than what is often claimed in media [57].
A typical US diet usually provides around 40–100 mg nitrate/day [58, 59] and 1-5 mg nitrite/day [60, 61]. On the basis of a conservative recommendation to consume 400 g of different fruits and vegetables per day at average nitrate concentrations, the dietary intake of nitrate would be 157 mg/day [30].
Ingested nitrate and nitrite is rapidly and completely be absorbed in the small intestine and taken up in the blood [62-65]. About 25% of blood nitrate is then absorbed by the salivary glands, of which 20% is converted to nitrite by commensal bacteria in the oral cavity [66, 67]. This nitrite is then swallowed and enters the blood circulation, where it becomes a precursor to NO generation via the nitrate-nitrite-NO pathway [37, 64].
Without this so called “entero-salivary circulation” of nitrate, which is dependent on oral bacteria, nitrate would leave the body unmodified as this chemically stable anion cannot be metabolized by mammalian enzymes. One can say that the entero-salivary circulation is a round-about-way of getting nitrite from nitrate, and up to 30% of the general population may not have the right oral bacteria to convert nitrate to nitrite [68] (which means they will not get any benefit from nitrate rich foods). Further, the necessary oral bacteria, when present, can easily be killed by anti-bacterial mouthwash [69]. Therefore it may be better to supplement with nitrite directly [70]. Nitrite can be seen as an NO donor, or storage form of NO (which decomposes in milliseconds after it has been liberated from its salt) [3, 29, 71], and is now at the forefront of NO biology [42]. However, warnings have been issued regarding supplementation with nitrite salts [72]. I will cover this debate on nitrite supplementation (commonly in the form of sodium nitrite) in a separate upcoming article…
Even though nitrate is less toxic than nitrite [73], very large amounts of nitrite (2000-3000 mg) needs to be ingested before toxicity sets in [68]. While veggies contain most nitrate and meat most nitrite, in this context intake of veggies should be prioritized because they also provide tons of other health promoting substances, which not only themselves confer health benefits, but also enhance the NO generation from dietary nitrite (regardless of whether this nitrite came from meat or veggies) [33].
Despite the beneficial effects of dietary nitrate and nitrite, conservative governmental institutions and regulations perceive that these are harmful dietary substances. The purported health risks of exposure to nitrite and nitrate are based on reports about met-hemoglobinemia in infants – “blue baby syndrome” – caused by drinks or food prepared with nitrate-rich and bacterially contaminated well water, as well as contaminated vegetables (such as spinach, celery, and carrots), occupational intoxication, increasing nitrate levels in soil and lakes as a result of fertilizer overuse, and the formation of potentially carcinogenic N-nitrosamines. Also, some epidemiological studies have shown a weak association between cured and processed meats, which contain nitrite and nitrate, and cancer [74].
This gave nitrite and nitrate a negative image, and as a result efforts have been made to remove as much nitrite and nitrate as possible from our drinking water and food. The Joint Food and Agricultural Organization/World Health Organization has set the Acceptable Daily Intake (ADI) for the nitrate at 1.7 mg/lb body weight (3.7 mg/kg) and for nitrite at 0.03 mg/lb body weight (0.06 mg/kg) [75]. Likewise, the Environmental Protection Agency (EPA) has set a daily Reference Dose for nitrate at 3.2 mg/lb body weight (7 mg/kg). This amounts to 222-420 mg nitrate and 3.6 mg nitrite for a 132 lb (60-kg) individual, which is below the doses that have been used in studies showing performance enhancing effects – around 500 mg nitrate [22] – as well as health promoting effects [7-10].
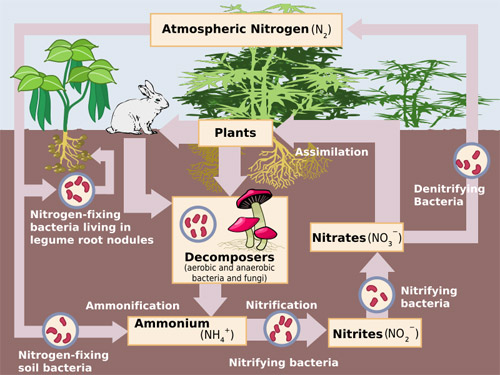
Figure 2: The nitrogen cycle - how nitrate and nitrite enters the natural ecosystem.
All the media scare about the purported hazards if nitrate and nitrite may make it appear as if dietary nitrate and nitrite are unnatural dangerous chemicals added by the industry. This is wrong; while cultivation and fertilization does add nitrate and nitrite to produce, vegetables also naturally contain nitrate and nitrite which enters the ecosystem via the nitrogen cycle, as illustrated in figure 2 [76]. Thus, dietary nitrate and nitrite are part of our natural diet, in the same way as are vitamins and minerals.
The weak and inconclusive data on the cancer risk of nitrite and nitrate [77, 78] are far outweighed by the well documented health benefits of supplementing the NO pool with dietary nitrate (and to a lesser extent nitrite). Nitrate has actually been shown to have beneficial health effects at intakes that traditionally have been considered to be toxic [30].
For example, the well-known health promoting and blood pressure-lowering DASH diet can provide over 1200 mg nitrate due to its high vegetable content, which exceeds the World Health Organization’s Acceptable Daily Intake for nitrate by 540% for a 132 lb (60-kg) adult. Further support comes from the traditional Japanese diet, which naturally provides 8.6 mg nitrate per lb body weight per day (1135 mg nitrate per day for a 132 lb individual) [46]. If nitrate really was that bad, the traditional Japanese diet wouldn’t be nourishing a population with the longest worldwide longevity.
These data call into question the rationale of recommendations to limit nitrate consumption from plant foods. Further, an effect of nitrite intake on cancer also seems less likely because significant amounts of nitrite are formed in the body. Fasting saliva contains 2 mg nitrite/L, and after consumption of an amount of nitrate equivalent to 7 oz of spinach (roughly 1400 mg nitrate), the nitrite concentration in saliva may rise as high as 72 mg/L [79]. That is much higher than the EPA standard for drinking water of 4.4 mg nitrite/L or the WHO Acceptable Daily Intake of 4.2 mg nitrite/d. Thus, the fuss and warnings about nitrate and nitrite need revision in the face of the undisputed health benefits of nitrate-enriched diets.
The potential health implications of increasing nitrate and nitrite intakes to fuel the nitrate-nitrite-NO pathway is critically important for health and function in older adults, in whom the arginine-eNOS-NO pathway is inefficient and/or dysfunctional [80-86].
Intake of nitrite is especially important in the elderly, who often have altered oral bacteria populations, dry mouth, and who take medications that affect stomach acidity and intestinal motility [87]. This is underscored by the finding that a large dose nitrate supplementation may be required to elevate blood levels of nitrate and nitrite in elderly, even among those who consume high-nitrate foods [87].
Aging is also characterized by endothelial dysfunction [83, 88], which is caused by disturbances in the arginine-eNOS-NO pathway and NO bioavailability, among other things [89-98]. The importance of nitrite in elderly adults is underscored by the fact that endothelial dysfunction is associated with reduced blood nitrite levels, and both reduced blood nitrite levels and endothelial dysfunction are correlated with an increasing numbers of cardiovascular risk factors [99, 100].
Thereby, an increased nitrite intake may provide a first line of defense against cardiovascular disease, especially in older people [3]. Moreover, the increased exercise tolerance seen after nitrate supplementation is greatly beneficial for elderly, who often cite exhaustion/fatigue as a critical barrier that prevents them from partaking in regular exercise [101, 102]. In this regard, nitrite/nitrate supplementation can indirectly help to prevent sarcopenia and frailty, which are deleterious age related conditions that primarily arise as a consequence from a sedentary lifestyle [103-106].
Several prominent scientists are actively reevaluating the health and performance effects of food sources of nitrates and nitrites [2, 11, 13, 15-17, 29-37, 43-47, 55, 64, 107-111]; hopefully these new results be reflected in upcoming regulatory and public health guidelines for dietary nitrite and nitrate exposures.
There is a major paradigm shift occurring in regards to the health and nutritional value of nitrate and nitrite. In the right context, they can provide enormous benefit to people in all walks of life. It has even been suggested that dietary nitrate and nitrite have a role in the diet as indispensable nutrients that many people are deficient in [30, 112].
Nitrate supplementation, by ramping up the nitrate-nitrite-NO pathway, has in most studies been shown to significantly increase performance and exercise tolerance, and reduce blood pressure and confer health promoting cardiovascular effects.
The nitrate-nitrite-NO pathway complements the better known L-arginine-NO pathway. One major difference is that production of NO from the nitrate-nitrite-NO pathway is greatly enhanced during hypoxia (low oxygen availability) and low pH, such as during exercise, which is when the L-arginine-NO pathway performs poorly. Thereby it kicks in when we need it the most.
What’s interesting is that the major supply of nitrate and nitrite in our bodies comes from our everyday diet. Thus, this alternative NO generating pathway can be harnessed therapeutically for prevention and treatment of diseases, while boosting performance and exercise capacity. Pretty good deal!
References:
1. Cosby, K., et al., Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nat Med, 2003. 9(12): p. 1498-505.
2. Lundberg, J.O. and E. Weitzberg, NO generation from nitrite and its role in vascular control. Arterioscler Thromb Vasc Biol, 2005. 25(5): p. 915-22.
3. Bryan, N.S., Nitrite in nitric oxide biology: cause or consequence? A systems-based review. Free Radic Biol Med, 2006. 41(5): p. 691-701.
4. van Faassen, E.E., et al., Nitrite as regulator of hypoxic signaling in mammalian physiology. Med Res Rev, 2009. 29(5): p. 683-741.
5. Feelisch, M., et al., Tissue processing of nitrite in hypoxia: an intricate interplay of nitric oxide-generating and -scavenging systems. J Biol Chem, 2008. 283(49): p. 33927-34.
6. Jones, A.M., Influence of dietary nitrate on the physiological determinants of exercise performance: a critical review. Appl Physiol Nutr Metab, 2014. 39(9): p. 1019-28.
7. Jones, A.M., Dietary nitrate supplementation and exercise performance. Sports Med, 2014. 44 Suppl 1: p. S35-45.
8. Lidder, S. and A.J. Webb, Vascular effects of dietary nitrate (as found in green leafy vegetables and beetroot) via the nitrate-nitrite-nitric oxide pathway. Br J Clin Pharmacol, 2013. 75(3): p. 677-96.
9. Machha, A. and A.N. Schechter, Dietary nitrite and nitrate: a review of potential mechanisms of cardiovascular benefits. Eur J Nutr, 2011. 50(5): p. 293-303.
10. Lara, J., et al., Effects of inorganic nitrate and beetroot supplementation on endothelial function: a systematic review and meta-analysis. Eur J Nutr, 2015.
11. Lundberg, J.O., E. Weitzberg, and M.T. Gladwin, The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat Rev Drug Discov, 2008. 7(2): p. 156-67.
12. Bryan, N.S., et al., Dietary nitrite restores NO homeostasis and is cardioprotective in endothelial nitric oxide synthase-deficient mice. Free Radic Biol Med, 2008. 45(4): p. 468-74.
13. Bailey, S.J., et al., Dietary nitrate supplementation reduces the O2 cost of low-intensity exercise and enhances tolerance to high-intensity exercise in humans. J Appl Physiol, 2009. 107(4): p. 1144-55.
14. Lansley, K.E., et al., Dietary nitrate supplementation reduces the O2 cost of walking and running: a placebo-controlled study. J Appl Physiol, 2011. 110(3): p. 591-600.
15. Larsen, F.J., et al., Dietary inorganic nitrate improves mitochondrial efficiency in humans. Cell Metab, 2011. 13(2): p. 149-59.
16. Bailey, S.J., et al., Dietary nitrate supplementation enhances muscle contractile efficiency during knee-extensor exercise in humans. J Appl Physiol, 2010. 109(1): p. 135-48.
17. Vanhatalo, A., et al., Dietary nitrate reduces muscle metabolic perturbation and improves exercise tolerance in hypoxia. J Physiol, 2011. 589(Pt 22): p. 5517-28.
18. Larsen, F.J., et al., Effects of dietary nitrate on oxygen cost during exercise. Acta Physiol (Oxf), 2007. 191(1): p. 59-66.
19. Coyle, E.F., Integration of the physiological factors determining endurance performance ability. Exerc Sport Sci Rev, 1995. 23: p. 25-63.
20. Lundberg, J.O., et al., Roles of dietary inorganic nitrate in cardiovascular health and disease. Cardiovasc Res, 2011. 89(3): p. 525-32.
21. Cermak, N.M., et al., No Improvement in Endurance Performance Following a Single Dose of Beetroot Juice. Int J Sport Nutr Exerc Metab, 2012.
22. Cermak, N.M., M.J. Gibala, and L.J. van Loon, Nitrate supplementation’s improvement of 10-km time-trial performance in trained cyclists. Int J Sport Nutr Exerc Metab, 2012. 22(1): p. 64-71.
23. Lansley, K.E., et al., Acute dietary nitrate supplementation improves cycling time trial performance. Med Sci Sports Exerc, 2011. 43(6): p. 1125-31.
24. Wilkerson, D.P., et al., Influence of acute dietary nitrate supplementation on 50 mile time trial performance in well-trained cyclists. Eur J Appl Physiol, 2012.
25. Bescos, R., et al., Sodium Nitrate Supplementation Does Not Enhance Performance of Endurance Athletes. Med Sci Sports Exerc, 2012.
26. Peacock, O., et al., Dietary Nitrate Does Not Enhance Running Performance in Elite Cross-country Skiers. Med Sci Sports Exerc, 2012.
27. Murphy, M., et al., Whole beetroot consumption acutely improves running performance. J Acad Nutr Diet, 2012. 112(4): p. 548-52.
28. Bond, H., L. Morton, and A.J. Braakhuis, Dietary nitrate supplementation improves rowing performance in well-trained rowers. Int J Sport Nutr Exerc Metab, 2012. 22(4): p. 251-6.
29. Dejam, A., et al., Emerging role of nitrite in human biology. Blood Cells Mol Dis, 2004. 32(3): p. 423-9.
30. Hord, N.G., Y. Tang, and N.S. Bryan, Food sources of nitrates and nitrites: the physiologic context for potential health benefits. Am J Clin Nutr, 2009. 90(1): p. 1-10.
31. Milkowski, A., et al., Nutritional epidemiology in the context of nitric oxide biology: a risk-benefit evaluation for dietary nitrite and nitrate. Nitric Oxide, 2010. 22(2): p. 110-9.
32. McKnight, G.M., et al., Dietary nitrate in man: friend or foe? Br J Nutr, 1999. 81(5): p. 349-58.
33. Lundberg, J.O., et al., Cardioprotective effects of vegetables: is nitrate the answer? Nitric Oxide, 2006. 15(4): p. 359-62.
34. Butler, A.R. and M. Feelisch, Therapeutic uses of inorganic nitrite and nitrate: from the past to the future. Circulation, 2008. 117(16): p. 2151-9.
35. Lundberg, J.O., et al., Nitrate, bacteria and human health. Nat Rev Microbiol, 2004. 2(7): p. 593-602.
36. Machha, A. and A.N. Schechter, Inorganic nitrate: a major player in the cardiovascular health benefits of vegetables? Nutr Rev, 2012. 70(6): p. 367-72.
37. Webb, A.J., et al., Acute blood pressure lowering, vasoprotective, and antiplatelet properties of dietary nitrate via bioconversion to nitrite. Hypertension, 2008. 51(3): p. 784-90.
38. Carlstrom, M., et al., Dietary inorganic nitrate reverses features of metabolic syndrome in endothelial nitric oxide synthase-deficient mice. Proc Natl Acad Sci U S A, 2010. 107(41): p. 17716-20.
39. Bryan, N.S., et al., Nitrite is a signaling molecule and regulator of gene expression in mammalian tissues. Nat Chem Biol, 2005. 1(5): p. 290-7.
40. Elrod, J.W., et al., Nitric oxide promotes distant organ protection: evidence for an endocrine role of nitric oxide. Proc Natl Acad Sci U S A, 2008. 105(32): p. 11430-5.
41. Schechter, A.N. and M.T. Gladwin, Hemoglobin and the paracrine and endocrine functions of nitric oxide. N Engl J Med, 2003. 348(15): p. 1483-5.
42. Gladwin, M.T., et al., The emerging biology of the nitrite anion. Nat Chem Biol, 2005. 1(6): p. 308-14.
43. Gilchrist, M., A.C. Shore, and N. Benjamin, Inorganic nitrate and nitrite and control of blood pressure. Cardiovasc Res, 2011. 89(3): p. 492-8.
44. Larsen, F.J., et al., Effects of dietary nitrate on blood pressure in healthy volunteers. N Engl J Med, 2006. 355(26): p. 2792-3.
45. Kapil, V., et al., Inorganic nitrate supplementation lowers blood pressure in humans: role for nitrite-derived NO. Hypertension, 2010. 56(2): p. 274-81.
46. Sobko, T., et al., Dietary nitrate in Japanese traditional foods lowers diastolic blood pressure in healthy volunteers. Nitric Oxide, 2010. 22(2): p. 136-40.
47. Vanhatalo, A., et al., Acute and chronic effects of dietary nitrate supplementation on blood pressure and the physiological responses to moderate-intensity and incremental exercise. Am J Physiol Regul Integr Comp Physiol, 2010. 299(4): p. R1121-31.
48. Carlstrom, M., et al., Dietary nitrate attenuates oxidative stress, prevents cardiac and renal injuries, and reduces blood pressure in salt-induced hypertension. Cardiovasc Res, 2011. 89(3): p. 574-85.
49. Kearney, P.M., et al., Global burden of hypertension: analysis of worldwide data. Lancet, 2005. 365(9455): p. 217-23.
50. He, J. and P.K. Whelton, Epidemiology and prevention of hypertension. Med Clin North Am, 1997. 81(5): p. 1077-97.
51. Zhang, Z., et al., Habitual coffee consumption and risk of hypertension: a systematic review and meta-analysis of prospective observational studies. Am J Clin Nutr, 2011. 93(6): p. 1212-9.
52. Qureshi, A.I., et al., Prevalence and trends of prehypertension and hypertension in United States: National Health and Nutrition Examination Surveys 1976 to 2000. Med Sci Monit, 2005. 11(9): p. CR403-9.
53. Yoon, S.S., Y. Ostchega, and T. Louis, Recent trends in the prevalence of high blood pressure and its treatment and control, 1999–2008, in Hyattsville, MD: US Department of Health and Human Services, CDC, National Center for Health Statistics; 2010, NCHS Data Brief no. 48. 2010.
54. Taddei, S., et al., Hypertension causes premature aging of endothelial function in humans. Hypertension, 1997. 29(3): p. 736-43.
55. Bryan, N.S., Food, Nutrition and the Nitric Oxide Pathway: Biochemistry and Bioactivity. 2009, Lancaster, PA: DesTech Publishing.
56. Stokes, K.Y., et al., Dietary nitrite prevents hypercholesterolemic microvascular inflammation and reverses endothelial dysfunction. Am J Physiol Heart Circ Physiol, 2009. 296(5): p. H1281-8.
57. Micha, R., S.K. Wallace, and D. Mozaffarian, Red and processed meat consumption and risk of incident coronary heart disease, stroke, and diabetes mellitus: a systematic review and meta-analysis. Circulation, 2010. 121(21): p. 2271-83.
58. Mensinga, T.T., G.J. Speijers, and J. Meulenbelt, Health implications of exposure to environmental nitrogenous compounds. Toxicol Rev, 2003. 22(1): p. 41-51.
59. Gangolli, S.D., et al., Nitrate, nitrite and N-nitroso compounds. Eur J Pharmacol, 1994. 292(1): p. 1-38.
60. Thomson, B., Nitrates and nitrites dietary exposure and risk assessment. 2004.
61. Dich, J., et al., Dietary intakes of nitrate, nitrite and NDMA in the Finnish Mobile Clinic Health Examination Survey. Food Addit Contam, 1996. 13(5): p. 541-52.
62. Florin, T.H., G. Neale, and J.H. Cummings, The effect of dietary nitrate on nitrate and nitrite excretion in man. Br J Nutr, 1990. 64(2): p. 387-97.
63. van Velzen, A.G., et al., The oral bioavailability of nitrate from nitrate-rich vegetables in humans. Toxicol Lett, 2008. 181(3): p. 177-81.
64. Lundberg, J.O. and M. Govoni, Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Radic Biol Med, 2004. 37(3): p. 395-400.
65. Hunault, C.C., et al., Bioavailability of sodium nitrite from an aqueous solution in healthy adults. Toxicol Lett, 2009. 190(1): p. 48-53.
66. Spiegelhalder, B., G. Eisenbrand, and R. Preussmann, Influence of dietary nitrate on nitrite content of human saliva: possible relevance to in vivo formation of N-nitroso compounds. Food Cosmet Toxicol, 1976. 14(6): p. 545-8.
67. Duncan, C., et al., Chemical generation of nitric oxide in the mouth from the enterosalivary circulation of dietary nitrate. Nat Med, 1995. 1(6): p. 546-51.
68. Bryan, N.S., Reduction of nitrate to nitrite by commensal oral bacteria, M. Mollica, Editor. 2012.
69. Govoni, M., et al., The increase in plasma nitrite after a dietary nitrate load is markedly attenuated by an antibacterial mouthwash. Nitric Oxide, 2008. 19(4): p. 333-7.
70. Sindler, A.L., et al., Inorganic nitrite supplementation for healthy arterial aging. J Appl Physiol (1985), 2014. 116(5): p. 463-77.
71. Gladwin, M.T., et al., Nitrite as a vascular endocrine nitric oxide reservoir that contributes to hypoxic signaling, cytoprotection, and vasodilation. Am J Physiol Heart Circ Physiol, 2006. 291(5): p. H2026-35.
72. Petroczi, A. and D.P. Naughton, Potentially fatal new trend in performance enhancement: a cautionary note on nitrite. J Int Soc Sports Nutr, 2010. 7: p. 25.
73. Lundberg, J.O., F.J. Larsen, and E. Weitzberg, Supplementation with nitrate and nitrite salts in exercise: a word of caution. J Appl Physiol, 2011. 111(2): p. 616-7.
74. Santarelli, R.L., F. Pierre, and D.E. Corpet, Processed meat and colorectal cancer: a review of epidemiologic and experimental evidence. Nutr Cancer, 2008. 60(2): p. 131-44.
75. EFS, A., Nitrate in vegetables: scientific opinion of the panel on contaminants in the food chain. The EFSA Journal, 2008. 689: p. 1-79.
76. Rudolf, M. and P.M. Kroneck, The nitrogen cycle: its biology. Met Ions Biol Syst, 2005. 43: p. 75-103.
77. van Loon, A.J., et al., Intake of nitrate and nitrite and the risk of gastric cancer: a prospective cohort study. Br J Cancer, 1998. 78(1): p. 129-35.
78. Powlson, D.S., et al., When does nitrate become a risk for humans? J Environ Qual, 2008. 37(2): p. 291-5.
79. Zetterquist, W., et al., Salivary contribution to exhaled nitric oxide. Eur Respir J, 1999. 13(2): p. 327-33.
80. Singh, N., et al., Ageing is associated with impairment of nitric oxide and prostanoid dilator pathways in the human forearm. Clin Sci (Lond), 2002. 102(5): p. 595-600.
81. Egashira, K., et al., Effects of age on endothelium-dependent vasodilation of resistance coronary artery by acetylcholine in humans. Circulation, 1993. 88(1): p. 77-81.
82. Lyons, D., et al., Impaired nitric oxide-mediated vasodilatation and total body nitric oxide production in healthy old age. Clin Sci (Lond), 1997. 93(6): p. 519-25.
83. Celermajer, D.S., et al., Aging is associated with endothelial dysfunction in healthy men years before the age-related decline in women. J Am Coll Cardiol, 1994. 24(2): p. 471-6.
84. Gerhard, M., et al., Aging progressively impairs endothelium-dependent vasodilation in forearm resistance vessels of humans. Hypertension, 1996. 27(4): p. 849-53.
85. Goubareva, I., et al., Age decreases nitric oxide synthesis and responsiveness in human platelets and increases formation of monocyte-platelet aggregates. Cardiovasc Res, 2007. 75(4): p. 793-802.
86. Casey, D.P., et al., Ageing reduces the compensatory vasodilatation during hypoxic exercise: the role of nitric oxide. J Physiol, 2011. 589(Pt 6): p. 1477-88.
87. Miller, G.D., et al., Plasma nitrate and nitrite are increased by a high-nitrate supplement but not by high-nitrate foods in older adults. Nutr Res, 2012. 32(3): p. 160-8.
88. Yavuz, B.B., et al., Advanced age is associated with endothelial dysfunction in healthy elderly subjects. Gerontology, 2008. 54(3): p. 153-6.
89. Vallance, P. and N. Chan, Endothelial function and nitric oxide: clinical relevance. Heart, 2001. 85(3): p. 342-50.
90. Smith, A.R. and T.M. Hagen, Vascular endothelial dysfunction in aging: loss of Akt-dependent endothelial nitric oxide synthase phosphorylation and partial restoration by (R)-alpha-lipoic acid. Biochem Soc Trans, 2003. 31(Pt 6): p. 1447-9.
91. Feletou, M. and P.M. Vanhoutte, Endothelial dysfunction: a multifaceted disorder (The Wiggers Award Lecture). Am J Physiol Heart Circ Physiol, 2006. 291(3): p. H985-1002.
92. Versari, D., et al., Endothelial dysfunction as a target for prevention of cardiovascular disease. Diabetes Care, 2009. 32 Suppl 2: p. S314-21.
93. Taddei, S., et al., Mechanisms of endothelial dysfunction: clinical significance and preventive non-pharmacological therapeutic strategies. Curr Pharm Des, 2003. 9(29): p. 2385-402.
94. Boger, R.H., The emerging role of asymmetric dimethylarginine as a novel cardiovascular risk factor. Cardiovasc Res, 2003. 59(4): p. 824-33.
95. Anderssohn, M., et al., Asymmetric dimethylarginine as a mediator of vascular dysfunction and a marker of cardiovascular disease and mortality: an intriguing interaction with diabetes mellitus. Diab Vasc Dis Res, 2010. 7(2): p. 105-18.
96. Boger, R.H., Asymmetric dimethylarginine, an endogenous inhibitor of nitric oxide synthase, explains the “L-arginine paradox” and acts as a novel cardiovascular risk factor. J Nutr, 2004. 134(10 Suppl): p. 2842S-2847S; discussion 2853S.
97. Ito, A., et al., Novel mechanism for endothelial dysfunction: dysregulation of dimethylarginine dimethylaminohydrolase. Circulation, 1999. 99(24): p. 3092-5.
98. Naseem, K.M., The role of nitric oxide in cardiovascular diseases. Mol Aspects Med, 2005. 26(1-2): p. 33-65.
99. Kleinbongard, P., et al., Plasma nitrite concentrations reflect the degree of endothelial dysfunction in humans. Free Radic Biol Med, 2006. 40(2): p. 295-302.
100. Brunner, H., et al., Endothelial function and dysfunction. Part II: Association with cardiovascular risk factors and diseases. A statement by the Working Group on Endothelins and Endothelial Factors of the European Society of Hypertension. J Hypertens, 2005. 23(2): p. 233-46.
101. Cooper, K.M., et al., Health barriers to walking for exercise in elderly primary care. Geriatr Nurs, 2001. 22(5): p. 258-62.
102. Schutzer, K.A. and B.S. Graves, Barriers and motivations to exercise in older adults. Prev Med, 2004. 39(5): p. 1056-61.
103. Hawkins, S.A., R.A. Wiswell, and T.J. Marcell, Exercise and the master athlete–a model of successful aging? J Gerontol A Biol Sci Med Sci, 2003. 58(11): p. 1009-11.
104. Marcell, T.J., Sarcopenia: causes, consequences, and preventions. J Gerontol A Biol Sci Med Sci, 2003. 58(10): p. M911-6.
105. Sayer, A.A., et al., The developmental origins of sarcopenia. J Nutr Health Aging, 2008. 12(7): p. 427-32.
106. Freiberger, E., C. Sieber, and K. Pfeifer, Physical activity, exercise, and sarcopenia – future challenges. Wien Med Wochenschr, 2011. 161(17-18): p. 416-25.
107. Archer, D.L., Evidence that ingested nitrate and nitrite are beneficial to health. J Food Prot, 2002. 65(5): p. 872-5.
108. Lundberg, J.O. and E. Weitzberg, NO generation from inorganic nitrate and nitrite: Role in physiology, nutrition and therapeutics. Arch Pharm Res, 2009. 32(8): p. 1119-26.
109. Tang, Y., H. Jiang, and N.S. Bryan, Nitrite and nitrate: cardiovascular risk-benefit and metabolic effect. Curr Opin Lipidol, 2011. 22(1): p. 11-5.
110. Bryan, N.S. and J. Loscalzo, Nitrite and Nitrate in Human Health and Disease. 2011: Humana Press.
111. Lundberg, J.O., Cardiovascular prevention by dietary nitrate and nitrite. Am J Physiol Heart Circ Physiol, 2009. 296(5): p. H1221-3.
112. Bryan, N.S., Cardioprotective actions of nitrite therapy and dietary considerations. Front Biosci, 2009. 14: p. 4793-808