Effects of 6-year Long-Term Testosterone Replacement Therapy (TRT) in Patients with ‘‘Diabesity’’

March 6th 2014 FDA approved Aveed for treatment of male hypogonadism, aka testosterone deficiency.[1] Aveed is a long-acting form of injectable testosterone called testosterone undecanoate. In Europe, testosterone undecanoate (under the name Nebido) has a long successful TRT track record for treatment of testosterone deficiency and its consequences (especially obesity, the metabolic syndrome and diabetes).[2-16]
In contrast to shorter acting forms of testosterone (e.g. cypionate), testosterone undecanoate only needs to be injected every 6 to 12 weeks, and thereby offers practical benefits to patients. (Comment: for Nebido (1000 mg per 4 ml) the initial interval is 6 weeks, followed by intervals of 10-14 weeks; for Aveed (750 mg per 3 ml) the initial interval is 4 weeks, followed by 10-week intervals).
Five days after the FDA approval a notable and impressive 6-year long TRT study was published, confirming the health benefits of TRT that have previously been found in shorter term studies... [44]
Background
The term “diabesity” was coined in the early 1970s to describe the occurrence of obesity and diabetes in the same individual.[17] Excessive amounts of body fat, especially abdominal (visceral) fat accumulation, is a well documented strong risk factor for insulin resistance and development of type-2 diabetes and cardiovascular disease.[18-26]
More and more studies are showing that testosterone deficiency contributes to development of obesity (both general and abdominal), insulin resistance, metabolic syndrome, type-2 diabetes and muscle loss. [27-32] In line with this, a growing number of intervention studies have demonstrated that TRT improves these outcomes.[2, 3][4, 28, 33-43]
Several recent 5-year long-term studies reported that TRT results in marked and significant weight (fat) loss, reduction in waist circumference and BMI, as well as improvements in blood cholesterol parameters and reductions in fasting glucose, HbA1c, CRP (in inflammatory marker) and liver enzymes.[3-5, 7]
Most recent long term study proving efficacy and safety of TRT use for 6-year
The longest study reported to date was conducted by a research group in Germany led by Dr Farid Saad, one of the most prominent worldwide leaders in the field of TRT research. This study was published March 11th, 2014 in the International Journal of Endocrinology.[44]
STUDY METHOD
Subjects were obese hypogonadal men with type-2 diabetes, aged between 41 and 73 years. All subjects had sub-normal total testosterone levels, mean 257 ng/dL (8.9 nmol/L), range 47-345 (1.63–11.79 nmol/L) and at least mild symptoms of hypogonadism assessed by the Aging Males’ Symptoms scale (AMS).
All men received treatment with testosterone undecanoate 1000 mg (Nebido, Bayer Pharma, Berlin, Germany), administered at baseline and at 6 weeks, and thereafter every 12 weeks for up to 72 months.
Waist circumference (WC) was measured midpoint between the iliac crest (top of the hip bone) and the lowest rib.
All 156 subjects were followed for at least one year, 146 for at least two years, 136 for three years, 114 for four years, 105 for five years, and 69 for six years.
RESULTS
Total Testosterone Levels during the 6-Year Period of Testosterone Treatment
Total testosterone levels showed a significant rise from 257 ng/dL (8.9 nmol/L) at the beginning of therapy to above 461 ng/dL (16 nmol/L) within the first year of therapy, and this physiological level remained constant throughout the course of treatment.
Waist Circumference
Figure 1 demonstrates the measured reduction in waist circumference subsequent to testosterone therapy in obese diabetic men. Waist circumference dropped from an average from 44.9 in (minimum 35 in, maximum 58 in) [114 cm (minimum 89 cm, maximum 148 cm)] to 40 in (minimum 32 in, maximum 47 in) [102 cm (minimum 82 cm, maximum 121 cm)] with a mean reduction of 4.6 in (11.56 cm) over the entire 6 year treatment period. The reduction in waist circumference was statistically significant at the end of each year compared to the previous year over the first five years, even at the end of six compared to five years.
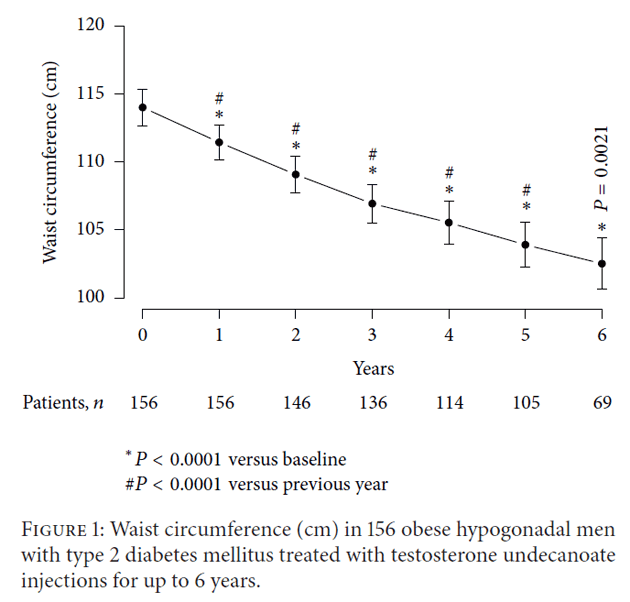
Weight Loss
Figure 2 shows the effects of TRT on body weight over the course of 6 years of therapy. Body weight decreased from an average of 249 lb (minimum 191 lb, maximum 311 lb) [113 kg (minimum 87 kg, maximum 141 kg) to 213 lb (minimum 176 lb, maximum 262 lb) [97 kg (min 80, max 119)] with a mean loss of 38.6 lb (17.5 kg) over the course of treatment. This decrease in body weight was statistically significant at the end of each year compared to the previous year over the first five years, even at the end of six compared to five years.
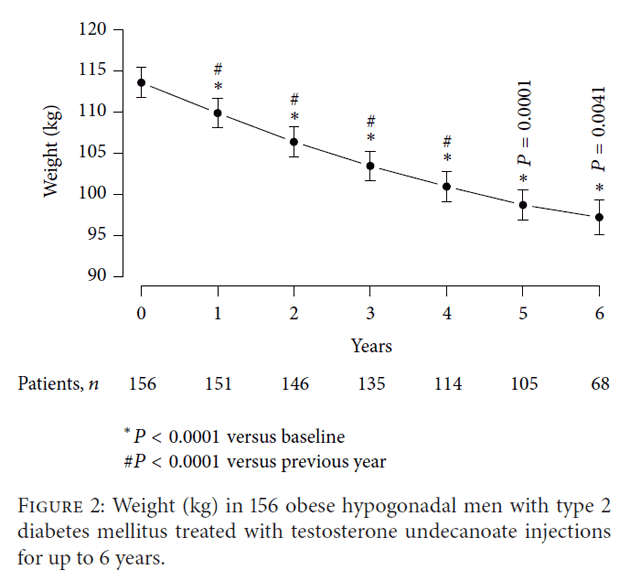
Percentage Change in Body Weight
A marked and significant decrease in percentage body weight was noted over the course of testosterone therapy. Over the entire 6-year observation period, patients lost 15% of their initial body weight (Figure 3). After one year, patients had lost 3.1% of their initial weight, after two years, 6.8%, after three years, 9.6%, after four years, 11.8%, after five years, 13.6%, and after 6 years, 15%. These successive yearly changes were statistically significant compared with baseline.
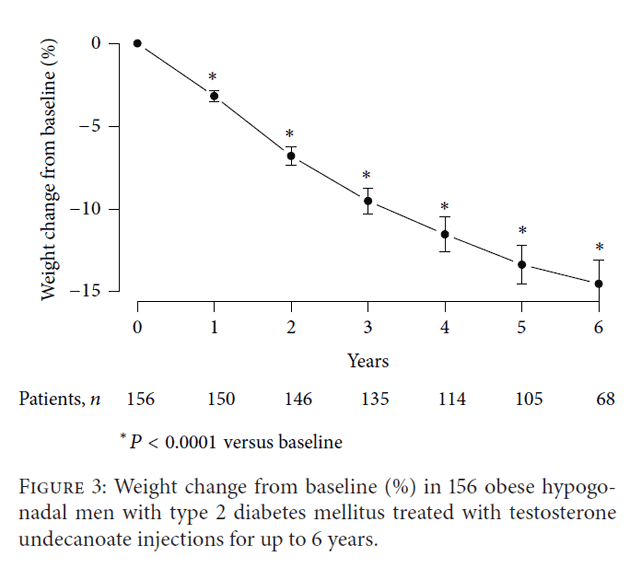
Blood Glucose Levels
There was a significant gradual decrease in fasting blood glucose from 128.37 ± 31.63 mg/dL (7.06 ± 1.74 mmol/L) to 101.55 ± 17.02 mg/dL (5.59 ± 0.94 mmol/). The decrease was significant after one year, further declined after two years (non-significant compared to level after year 1), and then reached a plateau with another slight but statistically significant decrease at five years compared to four years. Over the course of 6 year testosterone treatment, fasting blood glucose decreased by 27.14 ± 2.48 mg/dL (1.49 ± 0.14 mmol/L).
Hemoglobin A1c (HbA1c) Levels
The decrease in fasting blood glucose was accompanied by a marked decrease in HbA1c from 8.1% to 6.1% with a mean change of 1.9% at the end of the 6 year long testosterone treatment period.The decrease in HbA1c was progressive and statistically significant after one year, between two years and one year, between three and two years, between four and three years, and between five and four years and approached significance between six and five years.
Blood Cholesterol/Lipid Profiles Testosterone therapy improved lipid profiles as demonstrated with increase in high density lipoprotein cholesterol (HDL-C) by 35 ± 5%, significant reductions in total cholesterol by 32 ± 1.4%, low density lipoprotein cholesterol (LDL-C) by 25.9 ± 1.6%, and triglycerides (TG) by 30 ± 2%.
The mean changes in lipid profiles were gradual and progressive and were significant at each year when compared to baseline levels, reaching plateaus between three and four years. The ratio of total cholesterol to HDL cholesterol improved from 6 to 3. These changes reached a plateau after three years with further slight but not statistically significant decreases.
Blood Pressure, Inflammatory Status and Liver Function
In addition, testosterone treatment significantly reduced blood pressure, CRP (an inflammatory marker) and levels of the liver enzymes aspartate transaminase (AST) and alanine transaminase (ALT), suggesting a reduction in liver fat content, a reduced inflammatory response, and improvement in liver function.
Impact of Baseline Total Testosterone below versus above 231 ng/dL (8 nmol/L)
An analysis was made to see whether there were any differences in response among patients who at baseline had total testosterone levels below 231 ng/dL (8 nmol/L) versus those with levels between 231 ng/dL and 346 ng/dL (8 nmol/L and 12 nmol/L). It was found that obese diabetic men in the higher testosterone category responded to testosterone treatment equally well as those with lower baseline testosterone levels.
Safety
No adverse events were reported in patients treated with testosterone for up to 6 years.
CONCLUSION
The study authors concluded that long-term TRT for up to 6 years results in significant and sustained improvements in weight, type-2 diabetes, and other cardiometabolic risk factors in testosterone deficient obese, diabetic.[44] This study clearly demonstrates that TRT confers tremendous improvements in metabolic parameters and body composition, and may play an important role in the management/treatment of obesity and diabetes (diabesity) in men with testosterone deficiency.
Comments
Critics of TRT often state that there are no long term safety and efficacy studies on TRT. This 6-year long TRT study [44]and previous 5-years long studies [3, 4, 6, 7]blatantly refute those claims. Several of results from the above outlined 6-year long TRT study deserve special mention: [44]
First, this 6-year long study clearly demonstrates that TRT confers long term effects for the duration of treatment. Therefore, the response to a short term 3-month therapeutic trial of TRT, which has been suggested to be used as part of the diagnosis of hypogonadism [45], will not allow enough time for full expression of all the beneficial effects that have been documented in longer term TRT studies.[3-7, 44] For example, in the 6-year study outlined above, it took 1 year before significant and marked reductions were seen in waist circumference, weight, fasting glucose and HbA1c.[44]Further support for the importance of allowing enough treatment time comes from an analysis of the onset of effects of TRT and time span until maximum effects are achieved.[46]This analysis concluded that improvements in blood cholesterol/lipids and glycemic control may need up to 12 months for full expression.[46]Another important parameter when discussing TRT and time course of treatment is PSA (Prostate-Specific Antigen). PSA and prostate volume initially rises marginally, but it reaches a plateau at around 12 month (further increase should be related to aging rather than therapy).[46]This is a normal response and the initial PSA elevation should not be an indication to discontinue TRT too soon, if there are no contra-indications. Prostate growth is extremely sensitive to variations in androgen concentrations at very low concentrations, but becomes insensitive to changes in androgen concentrations at higher levels.[47]A short-term TRT treatment will not allow testosterone saturation of the prostate and PSA to the plateau.
Second, TRT with testosterone undecanoate 1000 mg at baseline, 6 wk and every 12 wk thereafter restored physiological testosterone levels within the first 12 months, and testosterone levels were maintained at this dosage regimen throughout the entire 6-year period. It should be noted that different testosterone preparations (e.g. testosterone cypionate, gel, pellet, oral) require different dosages and administration schedules in order to achieve a give target testosterone level.
Third, over the course of 6 years, TRT markedly and significantly reduced total cholesterol and LDL levels, and this reduction was very pronounced and sustained over the entire 6-year period of testosterone treatment. More importantly, TRT not only reduced the levels of total cholesterol and LDL, but also resulted in significant increases in HDL levels. Moreover, the ratio of total cholesterol to HDL cholesterol dropped from over 6 to below 3.5. As the total cholesterol to HDL ratio is a risk factor for cardiovascular disease [48, 49],this marked drop is notable.
Fourth, another important observation in this study was the marked and significant reduction in triglycerides (blood fats) in response to TRT. Since intra-abdominal (visceral fat) storage is driven by accumulation of triglycerides, this could help explain the reduction in waist circumference. In addition, the great reduction in triglycerides, especially when combined with the elevation in HDL, is an indication of an increased LDL particle size (i.e. a reduction in the atherogenic LDL fraction which is comprised of small LDL particles).[50]A decreased triglyceride/HDL ratio also indicates improved insulin sensitivity [50-54]. Thus the reduction in triglyceride/HDL ratio seen in the 6-year long testosterone replacement study [44]is in line with previous research showing that TRT improves insulin sensitivity in testosterone deficient men. [2, 55]
Fifth, the percent reduction in lipid levels (total cholesterol, LDL, and triglycerides) was close to 30%, which is a value similar to that attained by use of statins in men with abnormal blood lipids (dyslipidaemia).[56]
Sixth, it is notable that men with both very low and borderline low total testosterone levels responded equally well to this 6-year long-term testosterone treatment.
Finally, this study clearly shows that different outcomes plateau at different time points. For ex. the significant improvements in blood cholesterol and triglycerides (lipids) reached a plateau after 3-4 years of TRT, while the decrease in HbA1c was progressive and statistically significant up to five years of testosterone treatment. Importantly and interestingly, the reductions in waist circumference and body weight (and percentage change in body weight) were statistically significant at the end of each year compared to the previous year over the first five years, and even at the end of the sixth year compared to fifth year. This is an unusual and remarkable finding, as obesity interventions (both drugs and lifestyle changes) commonly are most effective the first year, and thereafter progressively lose their efficacy.[57-59]As this 6-year long study demonstrates, testosterone treatment excels in this regard. Let's hope in the near future this information will reach doctors working with patients who are sedentary, obese and suffer from cardio-metabolic complications.
Bottom Line
Did your doctor tell you that there isn't enough long-term safety and efficacy data to support the use of TRT? If so, do yourself a favor and get another doctor who is informed on the latest advances in testosterone research and men's health.
References:
1. FDA Approval History for Aveed (testosterone undecanoate). 2014.
2. Aversa, A., et al., Effects of testosterone undecanoate on cardiovascular risk factors and atherosclerosis in middle-aged men with late-onset hypogonadism and metabolic syndrome: results from a 24-month, randomized, double-blind, placebo-controlled study. J Sex Med, 2010. 7(10): p. 3495-503.
3. Saad, F., et al., Long-term treatment of hypogonadal men with testosterone produces substantial and sustained weight loss. Obesity (Silver Spring), 2013. 21(10): p. 1975-81.
4. Traish, A.M., et al., Long-term testosterone therapy in hypogonadal men ameliorates elements of the metabolic syndrome: an observational, long-term registry study. Int J Clin Pract, 2013.
5. Hackett, G., et al., Testosterone replacement therapy with long-acting testosterone undecanoate improves sexual function and quality-of-life parameters vs. placebo in a population of men with type 2 diabetes. J Sex Med, 2013. 10(6): p. 1612-27.
6. Francomano, D., et al., Effects of 5-year treatment with testosterone undecanoate on lower urinary tract symptoms in obese men with hypogonadism and metabolic syndrome. Urology, 2014. 83(1): p. 167-73.
7. Yassin, A. and G. Doros, Testosterone therapy in hypogonadal men results in sustained and clinically meaningful weight loss. Clin Obes, 2013. 3(3-4): p. 73-83.
8. Tan, W.S., et al., Efficacy and safety of long-acting intramuscular testosterone undecanoate in aging men: a randomised controlled study. BJU Int, 2013. 111(7): p. 1130-40.
9. Bouloux, P.M., et al., Effects of oral testosterone undecanoate therapy on bone mineral density and body composition in 322 aging men with symptomatic testosterone deficiency: a 1-year, randomized, placebo-controlled, dose-ranging study. Aging Male, 2013. 16(2): p. 38-47.
10. Zitzmann, M., et al., IPASS: a study on the tolerability and effectiveness of injectable testosterone undecanoate for the treatment of male hypogonadism in a worldwide sample of 1,438 men. J Sex Med, 2013. 10(2): p. 579-88.
11. Aversa, A., et al., Effects of long-acting testosterone undecanoate on bone mineral density in middle-aged men with late-onset hypogonadism and metabolic syndrome: results from a 36 months controlled study. Aging Male, 2012. 15(2): p. 96-102.
12. Haider, A., et al., A safety study of administration of parenteral testosterone undecanoate to elderly men over minimally 24 months. Andrologia, 2010. 42(6): p. 349-55.
13. Wang, C., et al., Pharmacokinetics and safety of long-acting testosterone undecanoate injections in hypogonadal men: an 84-week phase III clinical trial. J Androl, 2010. 31(5): p. 457-65.
14. Yassin, A.A. and F. Saad, Treatment of sexual dysfunction of hypogonadal patients with long-acting testosterone undecanoate (Nebido). World J Urol, 2006. 24(6): p. 639-44.
15. Moon du, G., et al., The efficacy and safety of testosterone undecanoate (Nebido((R))) in testosterone deficiency syndrome in Korean: a multicenter prospective study. J Sex Med, 2010. 7(6): p. 2253-60.
16. Moisey, R., J. Swinburne, and S. Orme, Serum testosterone and bioavailable testosterone correlate with age and body size in hypogonadal men treated with testosterone undecanoate (1000 mg IM--Nebido). Clin Endocrinol (Oxf), 2008. 69(4): p. 642-7.
17. Sims, E.A., et al., Endocrine and metabolic effects of experimental obesity in man. Recent Prog Horm Res, 1973. 29: p. 457-96.
18. Wannamethee, S.G., A.G. Shaper, and M. Walker, Overweight and obesity and weight change in middle aged men: impact on cardiovascular disease and diabetes. J Epidemiol Community Health, 2005. 59(2): p. 134-9.
19. Hamdy, O., S. Porramatikul, and E. Al-Ozairi, Metabolic obesity: the paradox between visceral and subcutaneous fat. Curr Diabetes Rev, 2006. 2(4): p. 367-73.
20. Despres, J.P., et al., Abdominal obesity and the metabolic syndrome: contribution to global cardiometabolic risk. Arterioscler Thromb Vasc Biol, 2008. 28(6): p. 1039-49.
21. Montague, C.T. and S. O'Rahilly, The perils of portliness: causes and consequences of visceral adiposity. Diabetes, 2000. 49(6): p. 883-8.
22. Huerta, J.M., et al., Risk of type 2 diabetes according to traditional and emerging anthropometric indices in Spain, a Mediterranean country with high prevalence of obesity: results from a large-scale prospective cohort study. BMC Endocr Disord, 2013. 13: p. 7.
23. Ashwell, M., P. Gunn, and S. Gibson, Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors: systematic review and meta-analysis. Obes Rev, 2012. 13(3): p. 275-86.
24. van Dijk, S.B., et al., Different anthropometric adiposity measures and their association with cardiovascular disease risk factors: a meta-analysis. Neth Heart J, 2012. 20(5): p. 208-18.
25. Kodama, S., et al., Comparisons of the strength of associations with future type 2 diabetes risk among anthropometric obesity indicators, including waist-to-height ratio: a meta-analysis. Am J Epidemiol, 2012. 176(11): p. 959-69.
26. Savva, S.C., D. Lamnisos, and A.G. Kafatos, Predicting cardiometabolic risk: waist-to-height ratio or BMI. A meta-analysis. Diabetes Metab Syndr Obes, 2013. 6: p. 403-419.
27. Roy, T.A., et al., Interrelationships of serum testosterone and free testosterone index with FFM and strength in aging men. Am J Physiol Endocrinol Metab, 2002. 283(2): p. E284-94.
28. Saad, F. and L.J. Gooren, The role of testosterone in the etiology and treatment of obesity, the metabolic syndrome, and diabetes mellitus type 2. J Obes, 2011. 2011.
29. Tang Fui, M.N., P. Dupuis, and M. Grossmann, Lowered testosterone in male obesity: Mechanisms, morbidity and management. Asian J Androl, 2013.
30. Yuki, A., et al., Relationship between low free testosterone levels and loss of muscle mass. Sci Rep, 2013. 3: p. 1818.
31. LeBlanc, E.S., et al., Higher testosterone levels are associated with less loss of lean body mass in older men. J Clin Endocrinol Metab, 2011. 96(12): p. 3855-63.
32. Katabami, T., et al., Serum free testosterone and metabolic syndrome in Japanese men. Endocr J, 2010. 57(6): p. 533-9.
33. Saad, F., Androgen therapy in men with testosterone deficiency: can testosterone reduce the risk of cardiovascular disease? Diabetes Metab Res Rev, 2012. 28 Suppl 2: p. 52-9.
34. Saad, F., et al., Testosterone as potential effective therapy in treatment of obesity in men with testosterone deficiency: a review. Curr Diabetes Rev, 2012. 8(2): p. 131-43.
35. Jones, T.H., et al., Testosterone replacement in hypogonadal men with type 2 diabetes and/or metabolic syndrome (the TIMES2 study). Diabetes Care, 2011. 34(4): p. 828-37.
36. Kenny, A.M., et al., Effects of transdermal testosterone on bone and muscle in older men with low bioavailable testosterone levels, low bone mass, and physical frailty. J Am Geriatr Soc, 2010. 58(6): p. 1134-43.
37. Cai, X., et al., Metabolic effects of testosterone replacement therapy on hypogonadal men with type 2 diabetes mellitus: a systematic review and meta-analysis of randomized controlled trials. Asian J Androl, 2014. 16(1): p. 146-52.
38. Kvorning, T., et al., Mechanical muscle function and lean body mass during supervised strength training and testosterone therapy in aging men with low-normal testosterone levels. J Am Geriatr Soc, 2013. 61(6): p. 957-62.
39. Storer, T.W., et al., Changes in muscle mass, muscle strength, and power but not physical function are related to testosterone dose in healthy older men. J Am Geriatr Soc, 2008. 56(11): p. 1991-9.
40. Sattler, F.R., et al., Testosterone and growth hormone improve body composition and muscle performance in older men. J Clin Endocrinol Metab, 2009. 94(6): p. 1991-2001.
41. Frederiksen, L., et al., Testosterone therapy increased muscle mass and lipid oxidation in aging men. Age (Dordr), 2012. 34(1): p. 145-56.
42. Boyanov, M.A., Z. Boneva, and V.G. Christov, Testosterone supplementation in men with type 2 diabetes, visceral obesity and partial androgen deficiency. Aging Male, 2003. 6(1): p. 1-7.
43. Kalinchenko, S.Y., et al., Effects of testosterone supplementation on markers of the metabolic syndrome and inflammation in hypogonadal men with the metabolic syndrome: the double-blinded placebo-controlled Moscow study. Clin Endocrinol (Oxf), 2010. 73(5): p. 602-12.
44. Haider, A., et al., Effects of Long-Term Testosterone Therapy on Patients with “Diabesity”: Results of Observational Studies of Pooled Analyses in Obese Hypogonadal Men with Type 2 Diabetes. International Journal of Endocrinology, 2014: p. Article ID 683515.
45. Black, A.M., A.G. Day, and A. Morales, The reliability of clinical and biochemical assessment in symptomatic late-onset hypogonadism: can a case be made for a 3-month therapeutic trial? BJU Int, 2004. 94(7): p. 1066-70.
46. Saad, F., et al., Onset of effects of testosterone treatment and time span until maximum effects are achieved. Eur J Endocrinol, 2011. 165(5): p. 675-85.
47. Morgentaler, A. and A.M. Traish, Shifting the paradigm of testosterone and prostate cancer: the saturation model and the limits of androgen-dependent growth. Eur Urol, 2009. 55(2): p. 310-20.
48. Kinosian, B., H. Glick, and G. Garland, Cholesterol and coronary heart disease: predicting risks by levels and ratios. Ann Intern Med, 1994. 121(9): p. 641-7.
49. Millan, J., et al., Lipoprotein ratios: Physiological significance and clinical usefulness in cardiovascular prevention. Vasc Health Risk Manag, 2009. 5: p. 757-65.
50. McLaughlin, T., et al., Is there a simple way to identify insulin-resistant individuals at increased risk of cardiovascular disease? Am J Cardiol, 2005. 96(3): p. 399-404.
51. Giannini, C., et al., The triglyceride-to-HDL cholesterol ratio: association with insulin resistance in obese youths of different ethnic backgrounds. Diabetes Care, 2011. 34(8): p. 1869-74.
52. Kannel, W.B., et al., Usefulness of the triglyceride-high-density lipoprotein versus the cholesterol-high-density lipoprotein ratio for predicting insulin resistance and cardiometabolic risk (from the Framingham Offspring Cohort). Am J Cardiol, 2008. 101(4): p. 497-501.
53. Li, C., et al., Does the association of the triglyceride to high-density lipoprotein cholesterol ratio with fasting serum insulin differ by race/ethnicity? Cardiovasc Diabetol, 2008. 7: p. 4.
54. Salazar, M.R., et al., Comparison of the abilities of the plasma triglyceride/high-density lipoprotein cholesterol ratio and the metabolic syndrome to identify insulin resistance. Diab Vasc Dis Res, 2013. 10(4): p. 346-52.
55. Kapoor, D., et al., Testosterone replacement therapy improves insulin resistance, glycaemic control, visceral adiposity and hypercholesterolaemia in hypogonadal men with type 2 diabetes. Eur J Endocrinol, 2006. 154(6): p. 899-906.
56. Mazza, F., et al., Effects of low-dose atorvastatin and rosuvastatin on plasma lipid profiles: a long-term, randomized, open-label study in patients with primary hypercholesterolemia. Am J Cardiovasc Drugs, 2008. 8(4): p. 265-70.
57. Sjostrom, L., et al., Randomised placebo-controlled trial of orlistat for weight loss and prevention of weight regain in obese patients. European Multicentre Orlistat Study Group. Lancet, 1998. 352(9123): p. 167-72.
58. Elfhag, K. and S. Rossner, Who succeeds in maintaining weight loss? A conceptual review of factors associated with weight loss maintenance and weight regain. Obes Rev, 2005. 6(1): p. 67-85.
59. Bray, G.A. and C. Bouchard, Handbook of Obesity, 4th edition2014: CRC Press.



