Combined Testosterone and GH therapy for best results on body composition and safety profiles

Many studies have highlighted the importance of investigating all major hormones, and correcting deficiencies and imbalances if present.[1-8] Given the known mechanisms of testosterone and GH/IGF-1 in building muscle (and possibly also DHEA in elderly) it is reasonable that age-related low levels of anabolic hormones contribute over time to sarcopenia and frailty.[1, 2, 4, 7, 9, 10]
Thus, multiple small effects in aggregate can lead to adverse loss of muscle and disability. In this scenario, if replacement was to occur, it would require lower doses of multiple anabolic hormones. An added benefit to this approach would be fewer side effects from the use of lower hormone doses [11]. In addition, multiple anabolic hormone replacement might also have beneficial additive or even synergistic effects.[11-13]
A notable study investigated whether supplementation with testosterone and GH together, in physiological doses, results in greater improvements in body composition and muscle performance in older men, compared to testosterone supplementation alone...
STUDY DESIGN AND PARTICIPANTS [13]
122 healthy overweight men (BMI 27) aged 66 to 74 years with baseline testosterone of 550 ng/dl or less, and IGF-I in lower adult range, 167 ng/dl or below, were randomized to receive transdermal testosterone 1% gel (5 g/day or 10 g/day, providing 5 mg or 10 mg testosterone/day) plus GH in doses of 0, 3, or 5 mcg/kg (per 2.2 lb) per day, for 16 wk.
MAIN OUTCOME MEASURES
Body composition by dual-energy x-ray absorptiometry, muscle performance, and safety tests.
RESULTS
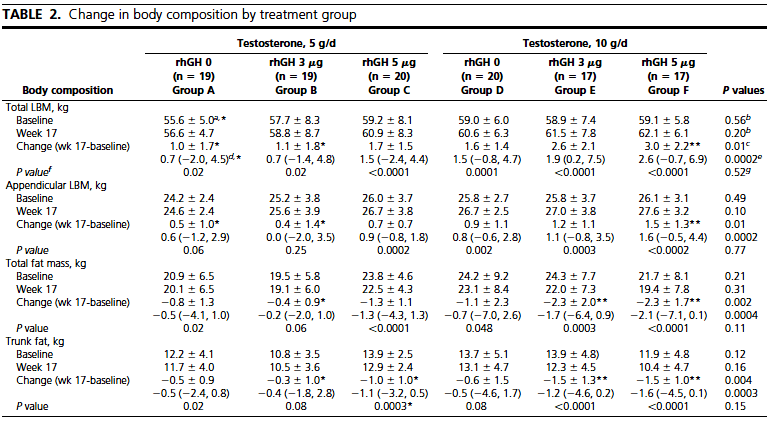
As indicated in the table, changes in body composition were dose-dependent across the six treatment groups:
 Reference: Sattler 2009 1 kg = 2.2 lb
Reference: Sattler 2009 1 kg = 2.2 lb
Total lean body mass increased by 1 - 3 kg (2.2 - 6.6 lb) and leg/arm (appendicular) lean body mass increased by 0.4 - 1.5 kg (0.9 - 3.3 kg).
Total fat mass decreased by 0.4 - 2.3 kg (0.9 - 5 lb), and trunk (abdominal) fat decreased by 0.5 - 1.5 kg (1.1 - 3.3 lb).
It should be noted that only testosterone 5g/day + GH 5 mcg/kg/day, and testosterone 10g/day + GH 3 mcg/kg/day and testosterone 10g/day + GH 5 mcg/kg/day (groups C, E and F) resulted in statistically significant results in all body composition variables.
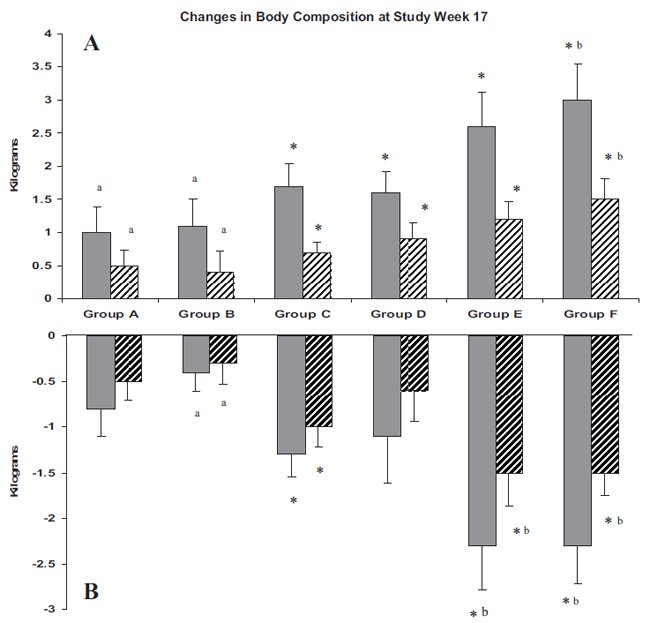
These body composition changes are illustrated graphically in the figure below:
Reference: Sattler 2009
As can be seen in the table and figure, the changes were clearly dose-dependent.
Maximum voluntary strength of upper and lower body muscles increased by 14 to 35 % (statistically significant only in the three highest dose groups), and correlated with changes in lag/arm lean mass. Aerobic endurance increased in all six groups.
In terms of metabolic effects, fasting blood sugar increased by 3 mg/dl across the entire study population but did not reach significance in any of the six groups. Indices of insulin resistance (HOMA-IR and QUICKI) were likewise unchanged in each of the six groups. Total and low-density lipoprotein (LDL) cholesterol didn't change in any of the six groups. High-density lipoprotein (HDL) cholesterol increased in most participants, but the increase was only significantly in group E by 4 mg/dl. Fasting triglycerides (blood fats) decreased on average by - 18 mg/dl, but the decrease was only significantly in group F by - 40 mg/dl.
SAFETY PARAMETERS
The testosterone + GH supplementation resulted in some predictable adverse events; however, they were modest and reversible.
Systolic and diastolic blood pressure increased similarly in each group with mean increases of 12 and 8 mm Hg, respectively. At follow-up over the ensuing 12 weeks after discontinuation of study therapies, the average increases in systolic and blood pressure were lower but still elevated by 9 and 6 mmHg, respectively.
Hematocrit increased significantly in four of the six groups; eight subjects had increases to 50–52%, one to 53%, but none to 54% or greater. After discontinuation of study interventions, hematocrit returned to less than50% in all subjects.
Although PSA increased in subjects by 0.2 ng/ml, it increased significantly only in group F from 1.1 to 1.8 ng/ml); no subject had a PSA increment greater than 1.4 ng/ml and values returned to baseline on repeated testing.
Conclusion and Comment
Supplemental testosterone produces significant gains in total and leg/arm lean mass, muscle strength, and aerobic endurance with significant reductions in whole-body and trunk fat. Results were further enhanced with GH supplementation, i.e. GH augments the beneficial effects of testosterone. It is impressive that these beneficial body composition results were achieved without any resistance training. One can imagine the further improvement of body composition by combining testosterone + GH therapy with resistance training.
The important take home message from these studies is that combined testosterone + GH therapy results in a larger magnitude of beneficial effects while keeping side effects at a minimum. Thus, testosterone combined with GH is a more effective muscle anabolic / fat loss treatment than either alone, and thus confers a better risk/benefit ratio.
It should be underscored that by combining testosterone and GH, a given anabolic and fat loss effect is achieved with a smaller dose of each compared with when testosterone and GH are given alone, and thus the risk for side effects is minimized. There may also be an extra effect that may not be achievable with either alone, which is likely because testosterone and GH/IGF-1 act on different metabolic pathways in an additive and possibly even synergistic manner.[11]
LEGAL NOTE
Ageless Forever does not offer multiple anabolic therapy for athletic enhancement or bodybuilding gains. This practice is considered illegal and unethical.
The studies we report on GH were conducted in subjects with established sub-optimal hormone levels.
Our goal at Ageless Forever is to replace (within the pysiological range) and balance sub-optimal hormone levels, as identified by comprehensive blood testing and clinical evaluation, for prevention of metabolic deterioration and health promotion.
References:
1. Bross, R., M. Javanbakht, and S. Bhasin, Anabolic interventions for aging-associated sarcopenia. J Clin Endocrinol Metab, 1999. 84(10): p. 3420-30.
2. Morley, J.E., Anabolic steroids and frailty. J Am Med Dir Assoc, 2010. 11(8): p. 533-6.
3. Villareal, D.T. and J.O. Holloszy, DHEA enhances effects of weight training on muscle mass and strength in elderly women and men. Am J Physiol Endocrinol Metab, 2006. 291(5): p. E1003-8.
4. McIntire, K.L. and A.R. Hoffman, The endocrine system and sarcopenia: potential therapeutic benefits. Curr Aging Sci, 2011. 4(3): p. 298-305.
5. Kovacheva, E.L., et al., Testosterone supplementation reverses sarcopenia in aging through regulation of myostatin, c-Jun NH2-terminal kinase, Notch, and Akt signaling pathways. Endocrinology, 2010. 151(2): p. 628-38.
6. Sayer, A.A., et al., New horizons in the pathogenesis, diagnosis and management of sarcopenia. Age Ageing, 2013. 42(2): p. 145-50.
7. Burks, T.N. and R.D. Cohn, One size may not fit all: anti-aging therapies and sarcopenia. Aging (Albany NY), 2011. 3(12): p. 1142-53.
8. Pasquali, R., et al., Sex-dependent role of glucocorticoids and androgens in the pathophysiology of human obesity. Int J Obes (Lond), 2008. 32(12): p. 1764-79.
9. Maggio, M., et al., The hormonal pathway to frailty in older men. J Endocrinol Invest, 2005. 28(11 Suppl Proceedings): p. 15-9.
10. Morley, J.E., M.J. Kim, and M.T. Haren, Frailty and hormones. Rev Endocr Metab Disord, 2005. 6(2): p. 101-8.
11. Giannoulis, M.G., et al., Hormone replacement therapy and physical function in healthy older men. Time to talk hormones? Endocr Rev, 2012. 33(3): p. 314-77.
12. Sattler, F., et al., Testosterone threshold levels and lean tissue mass targets needed to enhance skeletal muscle strength and function: the HORMA trial. J Gerontol A Biol Sci Med Sci, 2011. 66(1): p. 122-9.
13. Sattler, F.R., et al., Testosterone and growth hormone improve body composition and muscle performance in older men. J Clin Endocrinol Metab, 2009. 94(6): p. 1991-2001.



