The cardiovascular effects of testosterone and testosterone therapy are subject to intense investigation in medical research and have recently generated heated discussions among healthcare professionals.
While the main focus has been on testosterone per se, it is important to remember that testosterone is both a hormone in its own right, and a pro-hormone that gets converted to both estradiol and DHT (dihydrotestosterone). Estradiol and DHT exert effects themselves that are different from the effects of testosterone.
Therefore, when analyzing the effects of testosterone, especially supplemental testosterone administered as testosterone replacement therapy, it is critical to take into consideration how it affects downstream testosterone metabolites like estradiol and DHT.
Here I will present results from a recent systematic review and meta-analysis that specifically investigated how different routes of testosterone therapy administration (i.e different testosterone preparations) affect blood levels of testosterone and espcially DHT , and how this in turn relates to cardiovascular adverse events.[1]
Key Points
- The study presented here comprises two meta-analyses; one of cardiovascular adverse events in 35 randomized controlled trials (RCTs) of testosterone therapy lasting 12 weeks or more, and one of 32 studies reporting the effect of testosterone therapy on serum testosterone and dihydrotestosterone (DHT).
- The aim was to address cardiovascular effects as a function of the route of administration of testosterone therapy, possibly due to differential impact of testosterone preparations on elevations in DHT.
- No significant risk for cardiovascular adverse events was present when all testosterone therapy administration routes were grouped.
- No significant difference in the elevation of serum testosterone was present between intramuscular or transdermal testosterone preparations.
- Transdermal testosterone preparations elevated serum DHT (5.46-fold) to a greater magnitude than intramuscular testosterone preparations (2.20-fold).
- Too high serum DHT has been shown to be associated with cardiovascular risk in observational studies. The optimal range of DHT seems to be around 45-70 ng/dL.
- Differences in the degree to which serum DHT is elevated by different testosterone preparations and administration routes may explain possible differences in cardiovascular risk-to-benefit ratio.
What is known
Several previous longitudinal studies have shown that DHT is independently associated with incident cardiovascular disease [2], stroke [3], ischemic heart disease mortality [4] and all-cause mortality.[2, 4] All these prospective studies used the gold standard liquid chromatography-tandem mass spectrometry assay (LC-MS/MS) for hormone analyses.
The Cardiovascular Health Study, which included men aged 66–97 years old (mean age 76.5 years) most of whom (84%) rated their health as good to excellent, found that DHT has a nonlinear association with stroke risk; the lowest risk of stroke was at DHT levels of 50-75 ng/dL, with greater risk of stroke at DHT levels below 50 ng/dL or above 75 ng/dL.[3] Another report from the Cardiovascular Health Study demonstrated that DHT has curvilinear associations with incident cardiovascular disease and all-cause mortality in analyses adjusted for cardiovascular risk factors.[2] The lowest risk was seen at DHT levels of 50–74 ng/dL. Similarly to the association with stroke, greater risk for incident cardiovascular disease and all-cause mortality was seen at DHT levels below 50 ng/dL or above 74 ng/dL.[2] The follow-up time in these Cardiovascular Health Studies was 9-10 years.
The Health In Men Study (HIMS), a population-based cohort study of community-dwelling older men aged 70-89 years old, who were followed for approximately 7 years, found that having DHT levels in the third quartile at baseline, corresponding to 39-53 ng/dL (1.34–1.83 nmol/L), predicted reduced incidence of death from ischemic heart disease or any cause (all-cause mortality), regardless of age, overweight, or other risk factors, which were adjusted for in the analysis.[4] Total DHT and accurately calculated DHT had comparable and consistent associations with all-cause mortality. In this study, a threshold DHT level above 39 ng/dL (1.34 nmol/L) was unequivocally associated with lower ischemic heart disease mortality risk. Of note, this association was independent of SHBG levels.
What this study adds
The main finding in this meta-analysis of 35 eligible RCTs and more than 3,700 patients receiving testosterone therapy is that no significant increase in cardiovascular event risk was noted among studies of various testosterone therapy administration routes when analyzed together.[1] A second important finding is that oral and transdermal testosterone preparations cause greater DHT elevations than intramuscular administration.[1] A third finding was that a subgroup analysis found an increased cardiovascular risk with oral testosterone preparations, which raised DHT levels the most. This latter finding merits some important comments.
Oral testosterone preparations – data interpretation alert!
This meta-analysis only included four studies that used oral testosterone preparations.[5-8] One of the included studies was the notorious Copenhagen study [5] which was purely experimental and contributed most of the adverse effects; it was conducted in patients with alcoholic liver cirrhosis using a non-approved preparation of micronized oral testosterone that generated testosterone levels as high as 21,000 ng/dL (745 nmol/L), a value approximately 20 times the upper limit of the normal range.[5] It is not surprising that administering such a supraphysiological dose of a non-approved testosterone preparation to already diseased subjects would prove harmful. Therefore this study has no clinical relevance in the literature on physiological testosterone therapy in hypogonadal men and it is misleading to state that oral testosterone preparations increase cardiovascular risk.
The other three studies used oral testosterone undecanoate, and none of them reported any increase in cardiovascular risk, prostate changes or other adverse events.[6-8] Also, while the Copenhagen study generated a 9-fold elevation in DHT levels, the other three studies with oral testosterone undecanoate generated DHT elevations that were much lower, in the range of 1.8 to 3.8-fold.
Thus, the conclusion that oral testosterone causes the largest fold increase in DHT levels, and increases cardiovascular risk, is misleading and not valid.
Note about oral testosterone:
It should be noted that oral testosterone in general has a reputation for being hepatotoxic. However, this only applies to the old oral preparation methyltestosterone [9-12], which is being phased out from the market and replaced with the safe alternative, oral testosterone undecanoate.[11, 13, 14] A 10 year-long safety evaluation of oral testosterone undecanoate found no detrimental changes on neither liver, prostate nor cardiovascular parameters.[13]
Transdermal testosterone preparations
This meta-analysis found that transdermal testosterone preparations elevated serum DHT levels 5.5-fold, while intramuscular testosterone preparations delivered via injection elevated serum DHT levels only by 2.2-fold.[1] This is because transdermally administered testosterone is exposed to a high degree of 5-alpha reductase activity in the skin [15], which increases serum DHT levels more than does testosterone that is administered via injection. It should be underscored that passage of testosterone through the skin increases blood levels of DHT substantially.
Implications of elevated DHT for cardiovascular risk and mortality
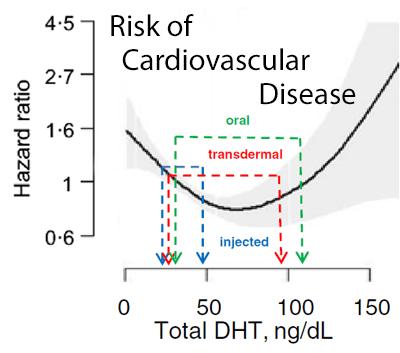
The larger elevation in DHT in relation to testosterone levels may have implications for cardiovascular risk. As noted above, the lowest risk of incident cardiovascular disease, stroke and all-cause mortality is seen at DHT levels in the range of 50–74 ng/dL [2, 3], while a level around 50 ng/dL has been associated with reduced incidence of death from ischemic heart disease.[4] This indicates that intramuscular testosterone preparations administered via injection elevate serum DHT levels into a range that is associated with reduced risk of cardiovascular disease and stroke, and reduced all-cause mortality. In contrast, transdermal testosterone preparations appear to elevate serum DHT into a higher range that may be associated with increased risk of incident cardiovascular disease, stroke and all-cause mortality. This is illustrated in figure 1. Due to inclusion of the non-relevant Copenhagen study, whose data grossly overwhelmed and distorted the interpretation of the oral testosterone group effects, the green lines in the figure should be disregarded.
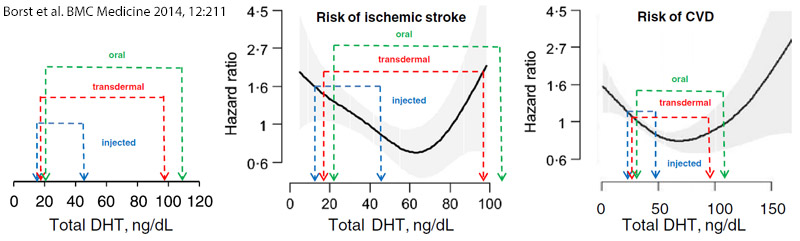
Figure 1: Comparison of DHT levels after testosterone treatment with DHT levels that are associated with cardiovascular disease risk.
* Left: Testosterone-induced elevation of DHT in the eight RCTs of testosterone injection, twenty RCTs of transdermal administration and four RCTs of oral testosterone administration, from meta-analysis.[1] Note the much larger elevation of serum DHT with transdermal testosterone administration compared to injected testosterone.
* Center: Data from panel 1 are overlaid on observational data from Shores et al. showing the relationship between serum DHT and 10 year risk of incident ischemic stroke in older men.[3] The solid line represents the estimated hazard ratio (HR) and the shaded area depicts the 95% confidence interval. All models are adjusted for age.
* Right: Data from panel 1 are overlaid on observational data from Shores et al. showing the relationship between serum DHT and incident cardiovascular disease risk. The solid line represents the estimated hazard ratio (HR) and the shaded area depicts the 95% confidence intervals. All models are adjusted for age.[2]
DHT, dihydrotestosterone; RCT, randomized controlled trial; CVD, cardiovascular disease
Conclusion
This meta-analysis underscores that there may be an optimal level of DHT, in the range of around 45-70 ng/dL, and shows that testosterone therapy with transdermal testosterone preparations cause a greater elevation in DHT levels than do injectable testosterone preparations.
Therefore, it is advisable for men who chose testosterone therapy via the transdermal route to have their DHT levels (in addition to testosterone and estradiol levels) regularly monitored.
Nevertheless, it should be underscored that other recent meta-analyses show that across all administration routes, there is no increased cardiovascular risk overall with testosterone replacement therapy that aims to restore physiological testosterone levels.[16, 17] Specifically, a recent meta-analysis performed on the largest number of studies collected so far, shows that testosterone replacement therapy is not related to any increase in cardiovascular risk, even when composite or single adverse events are considered. [16] To the contrary, in subjects with metabolic derangements, a protective effect of testosterone replacement therapy on cardiovascular risk is observed.[16] The grand conclusion from analyzing data of randomized controlled trials from the last 20 years is that testosterone replacement therapy in hypogonadal men is a valuable strategy in improving patient's metabolic profile, reducing body fat and increasing lean muscle mass, which would ultimately reduce the risk of heart disease.[16]
References:
1. Borst, S.E., et al., Cardiovascular risks and elevation of serum DHT vary by route of testosterone administration: a systematic review and meta-analysis. BMC Med, 2014. 12(1): p. 211.
2. Shores, M.M., et al., Testosterone, dihydrotestosterone, and incident cardiovascular disease and mortality in the cardiovascular health study. J Clin Endocrinol Metab, 2014. 99(6): p. 2061-8.
3. Shores, M.M., et al., Testosterone and dihydrotestosterone and incident ischaemic stroke in men in the Cardiovascular Health Study. Clin Endocrinol (Oxf), 2014. 81(5): p. 746-53.
4. Yeap, B.B., et al., In older men an optimal plasma testosterone is associated with reduced all-cause mortality and higher dihydrotestosterone with reduced ischemic heart disease mortality, while estradiol levels do not predict mortality. J Clin Endocrinol Metab, 2014. 99(1): p. E9-18.
5. The Copenhagen Study Group for Liver Diseases, Testosterone treatment of men with alcoholic cirrhosis: a double-blind study. Hepatology, 1986. 6(5): p. 807-13.
6. Legros, J.J., et al., Oral testosterone replacement in symptomatic late-onset hypogonadism: effects on rating scales and general safety in a randomized, placebo-controlled study. Eur J Endocrinol, 2009. 160(5): p. 821-31.
7. Emmelot-Vonk, M.H., et al., Effect of testosterone supplementation on functional mobility, cognition, and other parameters in older men: a randomized controlled trial. JAMA, 2008. 299(1): p. 39-52.
8. Chapman, I.M., et al., Effect of testosterone and a nutritional supplement, alone and in combination, on hospital admissions in undernourished older men and women. Am J Clin Nutr, 2009. 89(3): p. 880-9.
9. Werner, S.C., F.M. Hanger, and R.A. Kritzler, Jaundice during methyl testosterone therapy. Am J Med, 1950. 8(3): p. 325-31.
10. Glober, G.A. and J.A. Wilkerson, Biliary cirrhosis following the administration of methyltestosterone. JAMA, 1968. 204(2): p. 170-3.
11. Behre, H.M. and E. Nieschlag, Testosterone preparations for clinical use in males in Testosterone: Action, Deficiency, Substitution. 2014, Cambridge University Press. p. 309-335.
12. Westaby, D., et al., Liver damage from long-term methyltestosterone. Lancet, 1977. 2(8032): p. 262-3.
13. Gooren, L.J., A ten-year safety study of the oral androgen testosterone undecanoate. J Androl, 1994. 15(3): p. 212-5.
14. Wittert, G.A., et al., Oral testosterone supplementation increases muscle and decreases fat mass in healthy elderly males with low-normal gonadal status. J Gerontol A Biol Sci Med Sci, 2003. 58(7): p. 618-25.
15. Inui, S. and S. Itami, Androgen actions on the human hair follicle: perspectives. Exp Dermatol, 2013. 22(3): p. 168-71.
16. Corona, G., et al., Cardiovascular risk associated with testosterone-boosting medications: a systematic review and meta-analysis. Expert Opin Drug Saf, 2014. 13(10): p. 1327-51.
17. Corona, G., E. Maseroli, and M. Maggi, Injectable testosterone undecanoate for the treatment of hypogonadism. Expert Opin Pharmacother, 2014: p. 1-24.





