
In this article you will get critical information about the different niacin products and names, and what to look for when shopping niacin supplements…
The important thing to know about niacin is that it is available in a number of different formulations and products, which vary in time of release (absorption speed), efficacy (health promoting effects) and side effect profiles (flushing and liver strain). Also, the naming (terminology) of niacin products is not standardized and many names are used interchangeably, creating great confusion.[1] Here’s a handy overview:
Table: Overview of niacin products.[2]
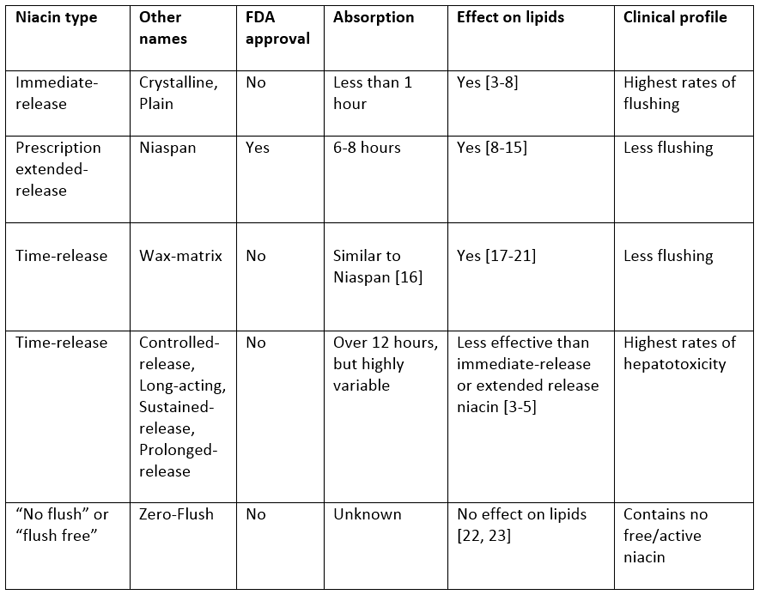
Immediate-release niacin
The landmark studies that demonstrated niacin’s impressive HDL boosting and Lp(a) reducing effects, as well as reduction in heart attacks, strokes and mortality, all used plain niacin .[24]
Immediate-release niacin is quickly absorbed and quickly excreted. Peak blood levels are obtained within 30 to 60 minutes of oral dosing and its half-life in the blood is approximately 1 hour.[25] Therefore it should be taken in multiple daily doses, after meals.
Flush-free niacin
Never take flush-free niacin! Flush-free niacin is inositol hexanicotinate, which contains six nicotinic acid (niacin) molecules bound to inositol. While niacin theoretically can be liberated from this formula - and product labels often prominently display the word “niacin” – it has been demonstrated that these products do not release any free niacin in the body, and thus are not bioavailable.[5, 23, 26] No wonder these products do not cause any flush! Studies have also showed that flush-free niacin products are “effect-free”.[22, 23] Despite this, flush-free niacin continues to be promoted as the ‘‘better tolerated’’ option for niacin therapy.
Therefore, one can make the argument that it is unethical to sell flush-free niacin supplements with health claims to improve blood lipids. People are wasting time and money on a worthless supplement and may be delaying appropriate treatment for a potentially serious health problem that would greatly benefit from the effective form of niacin, i.e. immediate-release niacin.
Time-release niacin
Time-release niacin - which is also known as sustained-release, controlled release, long-acting niacin or prolonged-release (there are probably other names for it as well) - has a greatly delayed absorption that typically exceeds 12 hours, and varies between brands.
Because of the greatly delayed absorption, time-release niacin causes less flushing but increases the risk of dose-related liver damaging (hepatotoxic) effects.[27] I will go into more detail on flushing and hepatotoxicity, as well as some other potential side effects of niacin - and what you can to do prevent them - in an upcoming article.
However, it should be highlighted that not all time-release niacin formulations are the same. Wax-matrix time-release niacin deserves special mention, more on that below.
Prescription extended-release niacin
Extended-release niacin has a moderately delayed absorption over 8 to 12 hours. Thus, its absorption is intermediate between immediate-release niacin and time-release niacin. This makes it suitable for once-daily bedtime dosing.
One extended-release niacin product called Niaspan is FDA approved for lipid/cholesterol treatment, and is only available by prescription. This may be an option for people who cannot tolerate the flush with immediate-release niacin.
Niacin formulations - confusing terminology
Most medical research uses the term extended-release niacin when referring to the prescription version of niacin, which is what I do in this article. However, sometimes the prescription version has been called prolonged-release.[28, 29] In daily parlance, prolonged-release niacin would likely refer to the non-prescription time-release form of niacin.
The United States Pharmacopeia (USP) is not of much help, as it does not make a specific distinction between the niacin formulation terminology. The only way to find out is to read the niacin product label. Look for immediate-release or wax-matrix niacin, which are the two non-prescription forms of niacin that are both effective and safe. If the label on a niacin product does not say immediate-release or wax-matrix anywhere, don’t buy it.
Efficacy comparison between niacin formulations
It is well documented that immediate-release niacin is more effective and safer than some time-release niacin.[3-5] When comparing immediate-release niacin with prescription extended-release niacin – as illustrated in figure 1 - immediate-release niacin has more beneficial effects on HDL and triglyceride levels, even at lower doses.[3] Both niacin formulations have similar dose–response effects on LDL levels.
Figure 1: Effects of immediate-release niacin vs. prescription extended release niacin on lipids.[3]
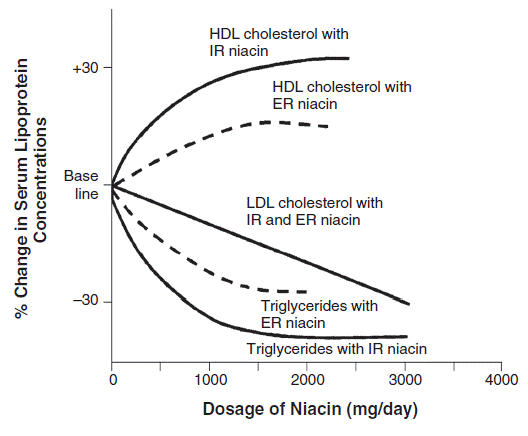
From Knopp RH. Drug treatment of lipid disorders. N. Engl. J. Med. 1999;341(7):498-511
IR = immediate-release niacin
ER = prescription extended-release niacin
Wax-matrix niacin - the best time-release niacin version!
When it comes to time-release niacin products, the first thing to know is that they all dose-dependently put some strain on the liver. [4, 30-35] This is because they have a longer dissolution/absorption rate than immediate-release niacin, and the liver strain is directly proportional to the dissolution/absorption rate.[35]
In doses that achieve equivalent improvements in blood lipid profiles, liver toxicity occurs more frequently with time-release niacin preparations than with immediate-release niacin.[36] Half of those who take time-release niacin develop elevations in liver transaminases.[31, 37] In contrast, immediate-release niacin in usual therapeutic doses (up to 3-4 g per day) almost never cause serious liver injury.[37, 38]
Nevertheless, it has been demonstrated that adverse liver effects with prescription extended-release niacin (Niaspan) up to 3 g per day are minor, and occur at rates similar to those reported for statin treatment.[12] Important to know, of all the over-the-counter time-release niacin formulations, wax-matrix niacin has the dissolution rate closes to and almost the same as the prescription extended-release niacin version.[16] Thus, the claim that prescription extended-release niacin has a unique release profile is not true.
Niaspan and Slo-Niacin (another non-prescription time-release version of niacin) are both polygel (a.k.a. methylcellulose) release products. The data on their dissolution (absorption) profiles are misleading because their dissolution studies were done in people taking them on an empty stomach (personal communication), when there is little peristaltic activity in the gut. When polygel time-release products are taken with food, the peristalsis of the stomach and bowel degrades the time-release complex and there is a significantly higher pulse release which causes flushing. Thus, taking Niaspan or Slo-Niacin with meals – which is often recommended in to reduce the flush intensity – may actually cause some “flush spikes”. Wax-matrix is not subject to degradation by peristalsis, and thus provides a more reliable release of niacin, even when taken after meals (as recommended).
Also, wax-matrix niacin is the only time-release form of niacin that has been proven in several studies to be effective and safe.[17-21] I will dedicate a separate upcoming article to the efficacy and safety of wax-matrix niacin.
It should be noted that wax-matrix niacin can be formulated with either canuba wax or rice bran wax. The niacin release rate is dictated by the manufacturing process, which is the same for the canuba and rice bran wax (they are produced by the same manufacturer). Thus, they release niacin at the same rate, and have the same efficacy and safety.
Summary
Niacin – aka vitamin B3 – is unquestionably a unique broad spectrum cholesterol improving “drug”. The important thing to know about niacin supplements is that there are several different versions. Never take the flush-free version of niacin, as it is effect-free and thus a waste of money.
With niacin products it is especially important to read the labels; look for a product that says immediate-release (or crystalline or plain) niacin. If you are looking for a time-release niacin, chose the one that lists the word “wax-matrix”.
Here’s a quick rule-of-thumb when starting niacin supplementation:
First try the immediate-release version of niacin to check your sensitivity to the flush. Build up to dose gradually from 100 mg twice daily for the first week, then double the daily dose each week until the target dose is reached, ideally 1500 to 3000 mg per day. Remember that with daily use, you will develop tolerance to the niacin flush after 1-2 weeks.
If you still are over-sensitive to the niacin flush – even after having developed tolerance – switch to wax-matrix time-release niacin; start taking 250 mg 3 times per day, and build up the dose to 750 mg 2-3 times per day.
In an upcoming article I will give detailed guidance on how to beat the niacin flush. Stay tuned…
In the meantime, check out these related niacin artcles:
References:
1. Knopp, R.H., Evaluating niacin in its various forms. Am J Cardiol, 2000. 86(12A): p. 51L-56L.
2. Villines, T.C., et al., Niacin: the evidence, clinical use, and future directions. Curr Atheroscler Rep, 2012. 14(1): p. 49-59.
3. Knopp, R.H., Drug treatment of lipid disorders. N Engl J Med, 1999. 341(7): p. 498-511.
4. McKenney, J.M., et al., A comparison of the efficacy and toxic effects of sustained- vs immediate-release niacin in hypercholesterolemic patients. JAMA, 1994. 271(9): p. 672-7.
5. Knopp, R.H., et al., Contrasting effects of unmodified and time-release forms of niacin on lipoproteins in hyperlipidemic subjects: clues to mechanism of action of niacin. Metabolism, 1985. 34(7): p. 642-50.
6. Clofibrate and niacin in coronary heart disease. JAMA, 1975. 231(4): p. 360-81.
7. Canner, P.L., et al., Fifteen year mortality in Coronary Drug Project patients: long-term benefit with niacin. J Am Coll Cardiol, 1986. 8(6): p. 1245-55.
8. Knopp, R.H., et al., Equivalent efficacy of a time-release form of niacin (Niaspan) given once-a-night versus plain niacin in the management of hyperlipidemia. Metabolism, 1998. 47(9): p. 1097-104.
9. Abbott Laboratories NIASPAN™ (niacin extended-release tablets) US prescribing information. Available at: http://www.niaspan.com 2005.
10. Capuzzi, D.M., et al., Efficacy and safety of an extended-release niacin (Niaspan): a long-term study. Am J Cardiol, 1998. 82(12A): p. 74U-81U; discussion 85U-86U.
11. Grundy, S.M., et al., Efficacy, safety, and tolerability of once-daily niacin for the treatment of dyslipidemia associated with type 2 diabetes: results of the assessment of diabetes control and evaluation of the efficacy of niaspan trial. Arch Intern Med, 2002. 162(14): p. 1568-76.
12. Guyton, J.R., et al., Effectiveness of once-nightly dosing of extended-release niacin alone and in combination for hypercholesterolemia. Am J Cardiol, 1998. 82(6): p. 737-43.
13. Morgan, J.M., et al., Effects of extended-release niacin on lipoprotein subclass distribution. Am J Cardiol, 2003. 91(12): p. 1432-6.
14. Morgan, J.M., D.M. Capuzzi, and J.R. Guyton, A new extended-release niacin (Niaspan): efficacy, tolerability, and safety in hypercholesterolemic patients. Am J Cardiol, 1998. 82(12A): p. 29U-34U; discussion 39U-41U.
15. Thoenes, M., et al., The effects of extended-release niacin on carotid intimal media thickness, endothelial function and inflammatory markers in patients with the metabolic syndrome. Int J Clin Pract, 2007. 61(11): p. 1942-8.
16. Poon, I.O., D.S. Chow, and D. Liang, Dissolution profiles of nonprescription extended-release niacin and inositol niacinate products. Am J Health Syst Pharm, 2006. 63(21): p. 2128-34.
17. Alderman, J.D., et al., Effect of a modified, well-tolerated niacin regimen on serum total cholesterol, high density lipoprotein cholesterol and the cholesterol to high density lipoprotein ratio. Am J Cardiol, 1989. 64(12): p. 725-9.
18. Keenan, J.M., et al., Niacin revisited. A randomized, controlled trial of wax-matrix sustained-release niacin in hypercholesterolemia. Arch Intern Med, 1991. 151(7): p. 1424-32.
19. Keenan, J.M., et al., A clinical trial of oat bran and niacin in the treatment of hyperlipidemia. J Fam Pract, 1992. 34(3): p. 313-9.
20. Keenan, J.M., et al., Treatment of hypercholesterolemia: comparison of younger versus older patients using wax-matrix sustained-release niacin. J Am Geriatr Soc, 1992. 40(1): p. 12-8.
21. Aronov, D.M., et al., Clinical trial of wax-matrix sustained-release niacin in a Russian population with hypercholesterolemia. Arch Fam Med, 1996. 5(10): p. 567-75.
22. Norris, R.B., "Flush-free niacin": dietary supplement may be "benefit-free". Prev Cardiol, 2006. 9(1): p. 64-5.
23. Keenan, J.M., Wax-matrix extended-release niacin vs inositol hexanicotinate: a comparison of wax-matrix, extended-release niacin to inositol hexanicotinate "no-flush" niacin in persons with mild to moderate dyslipidemia. J Clin Lipidol, 2013. 7(1): p. 14-23.
24. Carlson, L.A., Nicotinic acid: the broad-spectrum lipid drug. A 50th anniversary review. J Intern Med, 2005. 258(2): p. 94-114.
25. Piepho, R.W., The pharmacokinetics and pharmacodynamics of agents proven to raise high-density lipoprotein cholesterol. Am J Cardiol, 2000. 86(12A): p. 35L-40L.
26. Meyers, C.D., et al., Varying cost and free nicotinic acid content in over-the-counter niacin preparations for dyslipidemia. Ann Intern Med, 2003. 139(12): p. 996-1002.
27. Guyton, J.R. and H.E. Bays, Safety considerations with niacin therapy. Am J Cardiol, 2007. 99(6A): p. 22C-31C.
28. Carlson, L.A., Niaspan, the prolonged release preparation of nicotinic acid (niacin), the broad-spectrum lipid drug. Int J Clin Pract, 2004. 58(7): p. 706-13.
29. Vogt, A., et al., Prolonged-release nicotinic acid for the management of dyslipidemia: an update including results from the NAUTILUS study. Vasc Health Risk Manag, 2007. 3(4): p. 467-79.
30. Henkin, Y., K.C. Johnson, and J.P. Segrest, Rechallenge with crystalline niacin after drug-induced hepatitis from sustained-release niacin. JAMA, 1990. 264(2): p. 241-3.
31. Etchason, J.A., et al., Niacin-induced hepatitis: a potential side effect with low-dose time-release niacin. Mayo Clin Proc, 1991. 66(1): p. 23-8.
32. Lawrence, S.P., Transient focal hepatic defects related to sustained-release niacin. J Clin Gastroenterol, 1993. 16(3): p. 234-6.
33. Scheer, M.S., et al., Ultrasonographic findings in niacin-induced hepatitis. J Ultrasound Med, 1999. 18(4): p. 321-3.
34. Henkin, Y., et al., Niacin revisited: clinical observations on an important but underutilized drug. Am J Med, 1991. 91(3): p. 239-46.
35. McKenney, J., New perspectives on the use of niacin in the treatment of lipid disorders. Arch Intern Med, 2004. 164(7): p. 697-705.
36. Rader, J.I., R.J. Calvert, and J.N. Hathcock, Hepatic toxicity of unmodified and time-release preparations of niacin. Am J Med, 1992. 92(1): p. 77-81.
37. Pieper, J.A., Overview of niacin formulations: differences in pharmacokinetics, efficacy, and safety. Am J Health Syst Pharm, 2003. 60(13 Suppl 2): p. S9-14; quiz S25.
38. McKenney, J., Niacin for dyslipidemia: considerations in product selection. Am J Health Syst Pharm, 2003. 60(10): p. 995-1005.






