DHEA supplementation – specific health benefits for menopausal women

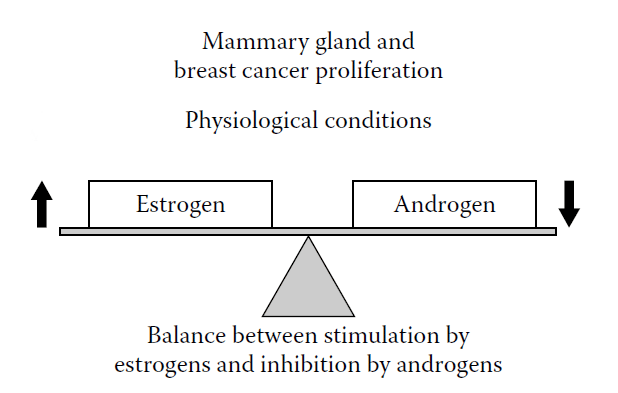
Breast cancer
Vagina
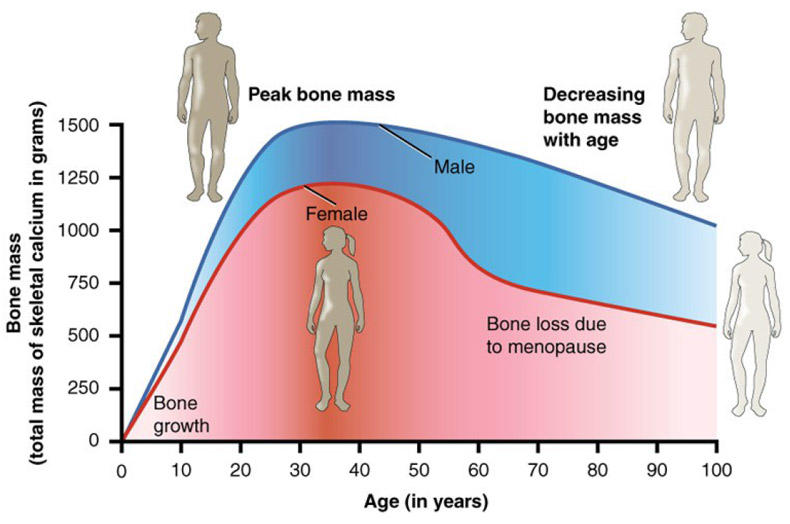
Bone
Cortisol balance
Skin and Wrinkles

Summary
1. Labrie, F., DHEA, important source of sex steroids in men and even more in women. Prog Brain Res, 2010. 182: p. 97-148.
2. Savineau, J.P., R. Marthan, and E. Dumas de la Roque, Role of DHEA in cardiovascular diseases. Biochem Pharmacol, 2013. 85(6): p. 718-26.
3. Shufelt, C., et al., DHEA-S levels and cardiovascular disease mortality in postmenopausal women: results from the National Institutes of Health--National Heart, Lung, and Blood Institute (NHLBI)-sponsored Women's Ischemia Syndrome Evaluation (WISE). J Clin Endocrinol Metab, 2010. 95(11): p. 4985-92.
4. Jimenez, M.C., et al., Low dehydroepiandrosterone sulfate is associated with increased risk of ischemic stroke among women. Stroke, 2013. 44(7): p. 1784-9.
5. Gordon, G.B., D.E. Bush, and H.F. Weisman, Reduction of atherosclerosis by administration of dehydroepiandrosterone. A study in the hypercholesterolemic New Zealand white rabbit with aortic intimal injury. J Clin Invest, 1988. 82(2): p. 712-20.
6. Arad, Y., et al., Dehydroepiandrosterone feeding prevents aortic fatty streak formation and cholesterol accumulation in cholesterol-fed rabbit. Arteriosclerosis, 1989. 9(2): p. 159-66.
7. Eich, D.M., et al., Inhibition of accelerated coronary atherosclerosis with dehydroepiandrosterone in the heterotopic rabbit model of cardiac transplantation. Circulation, 1993. 87(1): p. 261-9.
8. Yorek, M.A., et al., Effect of treatment of diabetic rats with dehydroepiandrosterone on vascular and neural function. Am J Physiol Endocrinol Metab, 2002. 283(5): p. E1067-75.
9. Liu, D. and J.S. Dillon, Dehydroepiandrosterone activates endothelial cell nitric-oxide synthase by a specific plasma membrane receptor coupled to Galpha(i2,3). J Biol Chem, 2002. 277(24): p. 21379-88.
10. Liu, D. and J.S. Dillon, Dehydroepiandrosterone stimulates nitric oxide release in vascular endothelial cells: evidence for a cell surface receptor. Steroids, 2004. 69(4): p. 279-89.
11. Simoncini, T., et al., Dehydroepiandrosterone modulates endothelial nitric oxide synthesis via direct genomic and nongenomic mechanisms. Endocrinology, 2003. 144(8): p. 3449-55.
12. Altman, R., et al., Inhibition of vascular inflammation by dehydroepiandrosterone sulfate in human aortic endothelial cells: roles of PPARalpha and NF-kappaB. Vascul Pharmacol, 2008. 48(2-3): p. 76-84.
13. Dillon, J.S., Dehydroepiandrosterone, dehydroepiandrosterone sulfate and related steroids: their role in inflammatory, allergic and immunological disorders. Curr Drug Targets Inflamm Allergy, 2005. 4(3): p. 377-85.
14. Chen, C.C. and C.R. Parker, Jr., Adrenal androgens and the immune system. Semin Reprod Med, 2004. 22(4): p. 369-77.
15. Gutierrez, G., et al., Dehydroepiandrosterone inhibits the TNF-alpha-induced inflammatory response in human umbilical vein endothelial cells. Atherosclerosis, 2007. 190(1): p. 90-9.
16. Khalil, A., et al., Dehydroepiandrosterone protects low density lipoproteins against peroxidation by free radicals produced by gamma-radiolysis of ethanol-water mixtures. Atherosclerosis, 1998. 136(1): p. 99-107.
17. Khalil, A., et al., Age-related decrease of dehydroepiandrosterone concentrations in low density lipoproteins and its role in the susceptibility of low density lipoproteins to lipid peroxidation. J Lipid Res, 2000. 41(10): p. 1552-61.
18. Cheng, H.H., X.J. Hu, and Q.R. Ruan, Dehydroepiandrosterone anti-atherogenesis effect is not via its conversion to estrogen. Acta Pharmacol Sin, 2009. 30(1): p. 42-53.
19. Jemal, A., et al., Cancer statistics, 2007. CA Cancer J Clin, 2007. 57(1): p. 43-66.
20. Walsh-Childers, K., H. Edwards, and S. Grobmyer, Covering women's greatest health fear: breast cancer information in consumer magazines. Health Commun, 2011. 26(3): p. 209-20.
21. Rossouw, J.E., et al., Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women's Health Initiative randomized controlled trial. JAMA, 2002. 288(3): p. 321-33.
22. Beral, V., Breast cancer and hormone-replacement therapy in the Million Women Study. Lancet, 2003. 362(9382): p. 419-27.
23. Beral, V., et al., Endometrial cancer and hormone-replacement therapy in the Million Women Study. Lancet, 2005. 365(9470): p. 1543-51.
24. Labrie, F., et al., Endocrine and intracrine sources of androgens in women: inhibition of breast cancer and other roles of androgens and their precursor dehydroepiandrosterone. Endocr Rev, 2003. 24(2): p. 152-82.
25. Kelloff, G.J., et al., New agents for cancer chemoprevention. J Cell Biochem Suppl, 1996. 26: p. 1-28.
26. Regelson, W. and M. Kalimi, Dehydroepiandrosterone (DHEA)--the multifunctional steroid. II. Effects on the CNS, cell proliferation, metabolic and vascular, clinical and other effects. Mechanism of action? Ann N Y Acad Sci, 1994. 719: p. 564-75.
27. Zumoff, B., et al., Abnormal 24-hr mean plasma concentrations of dehydroisoandrosterone and dehydroisoandrosterone sulfate in women with primary operable breast cancer. Cancer Res, 1981. 41(9 Pt 1): p. 3360-3.
28. Thomas, B.S., et al., Plasma dehydroepiandrosterone concentration in normal women and in patients with benign and malignant breast disease. Eur J Cancer, 1976. 12(5): p. 405-9.
29. Li, S., et al., Prevention by dehydroepiandrosterone of the development of mammary carcinoma induced by 7,12-dimethylbenz(a)anthracene (DMBA) in the rat. Breast Cancer Res Treat, 1994. 29(2): p. 203-17.
30. Gordon, G.B., L.M. Shantz, and P. Talalay, Modulation of growth, differentiation and carcinogenesis by dehydroepiandrosterone. Adv Enzyme Regul, 1987. 26: p. 355-82.
31. Schwartz, A.G., L. Pashko, and J.M. Whitcomb, Inhibition of tumor development by dehydroepiandrosterone and related steroids. Toxicol Pathol, 1986. 14(3): p. 357-62.
32. Shilkaitis, A., et al., Dehydroepiandrosterone inhibits the progression phase of mammary carcinogenesis by inducing cellular senescence via a p16-dependent but p53-independent mechanism. Breast Cancer Res, 2005. 7(6): p. R1132-40.
33. Hakkak, R., et al., Dehydroepiandrosterone intake protects against 7,12-dimethylbenz(a)anthracene-induced mammary tumor development in the obese Zucker rat model. Oncol Rep, 2010. 24(2): p. 357-62.
34. Dimitrakakis, C. and C. Bondy, Androgens and the breast. Breast Cancer Res, 2009. 11(5): p. 212.
35. Dimitrakakis, C., et al., A physiologic role for testosterone in limiting estrogenic stimulation of the breast. Menopause, 2003. 10(4): p. 292-8.
36. Hofling, M., et al., Testosterone inhibits estrogen/progestogen-induced breast cell proliferation in postmenopausal women. Menopause, 2007. 14(2): p. 183-90.
37. Zhou, J., et al., Testosterone inhibits estrogen-induced mammary epithelial proliferation and suppresses estrogen receptor expression. FASEB J, 2000. 14(12): p. 1725-30.
38. Glaser, R.L. and C. Dimitrakakis, Reduced breast cancer incidence in women treated with subcutaneous testosterone, or testosterone with anastrozole: a prospective, observational study. Maturitas, 2013. 76(4): p. 342-9.
39. Davis, S.R., et al., The effect of transdermal testosterone on mammographic density in postmenopausal women not receiving systemic estrogen therapy. J Clin Endocrinol Metab, 2009. 94(12): p. 4907-13.
40. Davis, S.R., et al., The incidence of invasive breast cancer among women prescribed testosterone for low libido. J Sex Med, 2009. 6(7): p. 1850-6.
41. Schwartz, A.G., Inhibition of spontaneous breast cancer formation in female C3H(Avy/a) mice by long-term treatment with dehydroepiandrosterone. Cancer Res, 1979. 39(3): p. 1129-32.
42. Glaser, R.L. and C. Dimitrakakis, Rapid response of breast cancer to neoadjuvant intramammary testosterone-anastrozole therapy: neoadjuvant hormone therapy in breast cancer. Menopause, 2014. 21(6): p. 673-8.
43. Labrie, F., et al., Metabolism of DHEA in postmenopausal women following percutaneous administration. J Steroid Biochem Mol Biol, 2007. 103(2): p. 178-88.
44. Stanczyk, F.Z., et al., Pharmacokinetics of dehydroepiandrosterone and its metabolites after long-term oral dehydroepiandrosterone treatment in postmenopausal women. Menopause, 2009. 16(2): p. 272-8.
45. Labrie, F., DHEA and Cancer Risk or Prevention?, in DHEA in Human Health and Aging, R.R. Watson, Editor. 2011, CRC Press. p. 159-179.
46. Schwartz, A.G. and L.L. Pashko, Dehydroepiandrosterone, glucose-6-phosphate dehydrogenase, and longevity. Ageing Res Rev, 2004. 3(2): p. 171-87.
47. Tian, W.N., et al., Importance of glucose-6-phosphate dehydrogenase activity for cell growth. J Biol Chem, 1998. 273(17): p. 10609-17.
48. Jiang, P., W. Du, and M. Wu, Regulation of the pentose phosphate pathway in cancer. Protein Cell, 2014.
49. Ho, H.Y., et al., Dehydroepiandrosterone induces growth arrest of hepatoma cells via alteration of mitochondrial gene expression and function. Int J Oncol, 2008. 33(5): p. 969-77.
50. Mac Bride, M.B., D.J. Rhodes, and L.T. Shuster, Vulvovaginal atrophy. Mayo Clin Proc, 2010. 85(1): p. 87-94.
51. Santoro, N. and J. Komi, Prevalence and impact of vaginal symptoms among postmenopausal women. J Sex Med, 2009. 6(8): p. 2133-42.
52. Levine, K.B., R.E. Williams, and K.E. Hartmann, Vulvovaginal atrophy is strongly associated with female sexual dysfunction among sexually active postmenopausal women. Menopause, 2008. 15(4 Pt 1): p. 661-6.
53. Tan, O., K. Bradshaw, and B.R. Carr, Management of vulvovaginal atrophy-related sexual dysfunction in postmenopausal women: an up-to-date review. Menopause, 2012. 19(1): p. 109-17.
54. North American Menopause, S., The role of local vaginal estrogen for treatment of vaginal atrophy in postmenopausal women: 2007 position statement of The North American Menopause Society. Menopause, 2007. 14(3 Pt 1): p. 355-69; quiz 370-1.
55. Barbaglia, G., et al., Trends in hormone therapy use before and after publication of the Women's Health Initiative trial: 10 years of follow-up. Menopause, 2009. 16(5): p. 1061-4.
56. Chism, L.A., Overcoming resistance and barriers to the use of local estrogen therapy for the treatment of vaginal atrophy. Int J Womens Health, 2012. 4: p. 551-7.
57. Labrie, F., et al., Intravaginal dehydroepiandrosterone (Prasterone), a physiological and highly efficient treatment of vaginal atrophy. Menopause, 2009. 16(5): p. 907-22.
58. Labrie, F., et al., Effect of intravaginal DHEA on serum DHEA and eleven of its metabolites in postmenopausal women. J Steroid Biochem Mol Biol, 2008. 111(3-5): p. 178-94.
59. Labrie, F., et al., Effect of 12-month dehydroepiandrosterone replacement therapy on bone, vagina, and endometrium in postmenopausal women. J Clin Endocrinol Metab, 1997. 82(10): p. 3498-505.
60. Panjari, M., et al., The safety of 52 weeks of oral DHEA therapy for postmenopausal women. Maturitas, 2009. 63(3): p. 240-5.
61. Stomati, M., et al., Six-month oral dehydroepiandrosterone supplementation in early and late postmenopause. Gynecol Endocrinol, 2000. 14(5): p. 342-63.
62. Bulun, S.E., et al., Regulation of aromatase expression in estrogen-responsive breast and uterine disease: from bench to treatment. Pharmacol Rev, 2005. 57(3): p. 359-83.
63. Baxendale, P.M., M.J. Reed, and V.H. James, Inability of human endometrium or myometrium to aromatize androstenedione. J Steroid Biochem, 1981. 14(3): p. 305-6.
64. Labrie, F., et al., Effect of intravaginal dehydroepiandrosterone (Prasterone) on libido and sexual dysfunction in postmenopausal women. Menopause, 2009. 16(5): p. 923-31.
65. Khosla, S., Pathogenesis of Osteoporosis. Transl Endocrinol Metab, 2010. 1(1): p. 55-86.
66. Jankowski, C.M., et al., Effects of dehydroepiandrosterone replacement therapy on bone mineral density in older adults: a randomized, controlled trial. J Clin Endocrinol Metab, 2006. 91(8): p. 2986-93.
67. Nair, K.S., et al., DHEA in elderly women and DHEA or testosterone in elderly men. N Engl J Med, 2006. 355(16): p. 1647-59.
68. von Muhlen, D., et al., Effect of dehydroepiandrosterone supplementation on bone mineral density, bone markers, and body composition in older adults: the DAWN trial. Osteoporos Int, 2008. 19(5): p. 699-707.
69. Weiss, E.P., et al., Dehydroepiandrosterone replacement therapy in older adults: 1- and 2-y effects on bone. Am J Clin Nutr, 2009. 89(5): p. 1459-67.
70. Harris, S.T., et al., Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. JAMA, 1999. 282(14): p. 1344-52.
71. de Villiers, T.J. and J.C. Stevenson, The WHI: the effect of hormone replacement therapy on fracture prevention. Climacteric, 2012. 15(3): p. 263-6.
72. Clarke, B.L. and S. Khosla, Androgens and bone. Steroids, 2009. 74(3): p. 296-305.
73. Manolagas, S.C., C.A. O'Brien, and M. Almeida, The role of estrogen and androgen receptors in bone health and disease. Nat Rev Endocrinol, 2013. 9(12): p. 699-712.
74. Davis, S.R., et al., Testosterone enhances estradiol's effects on postmenopausal bone density and sexuality. Maturitas, 1995. 21(3): p. 227-36.
75. Raisz, L.G., et al., Comparison of the effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab, 1996. 81(1): p. 37-43.
76. Coxam, V., et al., Effects of dihydrotestosterone alone and combined with estrogen on bone mineral density, bone growth, and formation rates in ovariectomized rats. Bone, 1996. 19(2): p. 107-14.
77. Genazzani, A.D., et al., Oral dehydroepiandrosterone supplementation modulates spontaneous and growth hormone-releasing hormone-induced growth hormone and insulin-like growth factor-1 secretion in early and late postmenopausal women. Fertil Steril, 2001. 76(2): p. 241-8.
78. Morales, A.J., et al., The effect of six months treatment with a 100 mg daily dose of dehydroepiandrosterone (DHEA) on circulating sex steroids, body composition and muscle strength in age-advanced men and women. Clin Endocrinol (Oxf), 1998. 49(4): p. 421-32.
79. Morales, A.J., et al., Effects of replacement dose of dehydroepiandrosterone in men and women of advancing age. J Clin Endocrinol Metab, 1994. 78(6): p. 1360-7.
80. Villareal, D.T., J.O. Holloszy, and W.M. Kohrt, Effects of DHEA replacement on bone mineral density and body composition in elderly women and men. Clin Endocrinol (Oxf), 2000. 53(5): p. 561-8.
81. Jankowski, C.M., et al., Increases in bone mineral density in response to oral dehydroepiandrosterone replacement in older adults appear to be mediated by serum estrogens. J Clin Endocrinol Metab, 2008. 93(12): p. 4767-73.
82. Locatelli, V. and V.E. Bianchi, Effect of GH/IGF-1 on Bone Metabolism and Osteoporsosis. Int J Endocrinol, 2014. 2014: p. 235060.
83. Giustina, A., G. Mazziotti, and E. Canalis, Growth hormone, insulin-like growth factors, and the skeleton. Endocr Rev, 2008. 29(5): p. 535-59.
84. Niemann, I., et al., The association between insulin-like growth factor I and bone turnover markers in the general adult population. Bone, 2013. 56(1): p. 184-90.
85. Callewaert, F., et al., Skeletal sexual dimorphism: relative contribution of sex steroids, GH-IGF1, and mechanical loading. J Endocrinol, 2010. 207(2): p. 127-34.
86. Woods, N.F., et al., Increased urinary cortisol levels during the menopausal transition. Menopause, 2006. 13(2): p. 212-21.
87. Woods, N.F., E.S. Mitchell, and K. Smith-Dijulio, Cortisol levels during the menopausal transition and early postmenopause: observations from the Seattle Midlife Women's Health Study. Menopause, 2009. 16(4): p. 708-18.
88. Meldrum, D.R., et al., Pituitary hormones during the menopausal hot flash. Obstet Gynecol, 1984. 64(6): p. 752-6.
89. Cagnacci, A., et al., Increased cortisol level: a possible link between climacteric symptoms and cardiovascular risk factors. Menopause, 2011. 18(3): p. 273-8.
90. Phillips, A.C., et al., Cortisol, DHEA sulphate, their ratio, and all-cause and cause-specific mortality in the Vietnam Experience Study. Eur J Endocrinol, 2010. 163(2): p. 285-92.
91. Phillips, A.C., et al., Cortisol, DHEAS, their ratio and the metabolic syndrome: evidence from the Vietnam Experience Study. Eur J Endocrinol, 2010. 162(5): p. 919-23.
92. Kalmijn, S., et al., A prospective study on cortisol, dehydroepiandrosterone sulfate, and cognitive function in the elderly. J Clin Endocrinol Metab, 1998. 83(10): p. 3487-92.
93. Lupien, S.J., et al., Cortisol levels during human aging predict hippocampal atrophy and memory deficits. Nat Neurosci, 1998. 1(1): p. 69-73.
94. Hadoke, P.W., J. Iqbal, and B.R. Walker, Therapeutic manipulation of glucocorticoid metabolism in cardiovascular disease. Br J Pharmacol, 2009. 156(5): p. 689-712.
95. Whitworth, J.A., et al., Cardiovascular consequences of cortisol excess. Vasc Health Risk Manag, 2005. 1(4): p. 291-9.
96. Duclos, M., et al., Increased cortisol bioavailability, abdominal obesity, and the metabolic syndrome in obese women. Obes Res, 2005. 13(7): p. 1157-66.
97. Wallerius, S., et al., Rise in morning saliva cortisol is associated with abdominal obesity in men: a preliminary report. J Endocrinol Invest, 2003. 26(7): p. 616-9.
98. Poehlman, E.T., Menopause, energy expenditure, and body composition. Acta Obstet Gynecol Scand, 2002. 81(7): p. 603-11.
99. Toth, M.J., et al., Menopause-related changes in body fat distribution. Ann N Y Acad Sci, 2000. 904: p. 502-6.
100. Shi, H. and D.J. Clegg, Sex differences in the regulation of body weight. Physiol Behav, 2009. 97(2): p. 199-204.
101. Genazzani, A.R., et al., Long-term low-dose oral administration of dehydroepiandrosterone modulates adrenal response to adrenocorticotropic hormone in early and late postmenopausal women. Gynecol Endocrinol, 2006. 22(11): p. 627-35.
102. Kroboth, P.D., et al., Influence of DHEA administration on 24-hour cortisol concentrations. J Clin Psychopharmacol, 2003. 23(1): p. 96-9.
103. Alhaj, H.A., A.E. Massey, and R.H. McAllister-Williams, Effects of DHEA administration on episodic memory, cortisol and mood in healthy young men: a double-blind, placebo-controlled study. Psychopharmacology (Berl), 2006. 188(4): p. 541-51.
104. McQuade, R. and A.H. Young, Future therapeutic targets in mood disorders: the glucocorticoid receptor. Br J Psychiatry, 2000. 177: p. 390-5.
105. Otte, C., et al., A meta-analysis of cortisol response to challenge in human aging: importance of gender. Psychoneuroendocrinology, 2005. 30(1): p. 80-91.
106. Stein-Behrens, B.A. and R.M. Sapolsky, Stress, glucocorticoids, and aging. Aging (Milano), 1992. 4(3): p. 197-210.
107. Apostolova, G., et al., Dehydroepiandrosterone inhibits the amplification of glucocorticoid action in adipose tissue. Am J Physiol Endocrinol Metab, 2005. 288(5): p. E957-64.
108. Tagawa, N., et al., Alternative mechanism for anti-obesity effect of dehydroepiandrosterone: possible contribution of 11beta-hydroxysteroid dehydrogenase type 1 inhibition in rodent adipose tissue. Steroids, 2011. 76(14): p. 1546-53.
109. McNelis, J.C., et al., Dehydroepiandrosterone exerts antiglucocorticoid action on human preadipocyte proliferation, differentiation, and glucose uptake. Am J Physiol Endocrinol Metab, 2013. 305(9): p. E1134-44.
110. Rask, E., et al., Tissue-specific dysregulation of cortisol metabolism in human obesity. J Clin Endocrinol Metab, 2001. 86(3): p. 1418-21.
111. Rask, E., et al., Tissue-specific changes in peripheral cortisol metabolism in obese women: increased adipose 11beta-hydroxysteroid dehydrogenase type 1 activity. J Clin Endocrinol Metab, 2002. 87(7): p. 3330-6.
112. Libe, R., et al., Effects of dehydroepiandrosterone (DHEA) supplementation on hormonal, metabolic and behavioral status in patients with hypoadrenalism. J Endocrinol Invest, 2004. 27(8): p. 736-41.
113. Villareal, D.T. and J.O. Holloszy, Effect of DHEA on abdominal fat and insulin action in elderly women and men: a randomized controlled trial. JAMA, 2004. 292(18): p. 2243-8.
114. Tan, C.Y., et al., Skin thickness measurement by pulsed ultrasound: its reproducibility, validation and variability. Br J Dermatol, 1982. 106(6): p. 657-67.
115. Henry, F., et al., Age-related changes in facial skin contours and rheology. J Am Geriatr Soc, 1997. 45(2): p. 220-2.
116. Fisher, G.J., et al., Mechanisms of photoaging and chronological skin aging. Arch Dermatol, 2002. 138(11): p. 1462-70.
117. Brincat, M., et al., Skin collagen changes in postmenopausal women receiving different regimens of estrogen therapy. Obstet Gynecol, 1987. 70(1): p. 123-7.
118. Affinito, P., et al., Effects of postmenopausal hypoestrogenism on skin collagen. Maturitas, 1999. 33(3): p. 239-47.
119. Brincat, M., et al., A study of the decrease of skin collagen content, skin thickness, and bone mass in the postmenopausal woman. Obstet Gynecol, 1987. 70(6): p. 840-5.
120. Delaney, M.F., Strategies for the prevention and treatment of osteoporosis during early postmenopause. Am J Obstet Gynecol, 2006. 194(2 Suppl): p. S12-23.
121. McClung, M.R., The menopause and HRT. Prevention and management of osteoporosis. Best Pract Res Clin Endocrinol Metab, 2003. 17(1): p. 53-71.
122. Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause, 2010. 17(1): p. 25-54; quiz 55-6.
123. Brincat, M., et al., Long-term effects of the menopause and sex hormones on skin thickness. Br J Obstet Gynaecol, 1985. 92(3): p. 256-9.
124. Pierard, G.E., et al., Effect of hormone replacement therapy for menopause on the mechanical properties of skin. J Am Geriatr Soc, 1995. 43(6): p. 662-5.
125. Makrantonaki, E. and C.C. Zouboulis, Androgens and ageing of the skin. Curr Opin Endocrinol Diabetes Obes, 2009. 16(3): p. 240-5.
126. Savvas, M., et al., Type III collagen content in the skin of postmenopausal women receiving oestradiol and testosterone implants. Br J Obstet Gynaecol, 1993. 100(2): p. 154-6.
127. Pierard, G.E., et al., Revisiting the cutaneous impact of oral hormone replacement therapy. Biomed Res Int, 2013. 2013: p. 971760.
128. Zouboulis, C.C., et al., Sexual hormones in human skin. Horm Metab Res, 2007. 39(2): p. 85-95.
129. Chen, W., et al., Testosterone synthesized in cultured human SZ95 sebocytes derives mainly from dehydroepiandrosterone. Exp Dermatol, 2010. 19(5): p. 470-2.
130. Fritsch, M., C.E. Orfanos, and C.C. Zouboulis, Sebocytes are the key regulators of androgen homeostasis in human skin. J Invest Dermatol, 2001. 116(5): p. 793-800.
131. Chen, W., D. Thiboutot, and C.C. Zouboulis, Cutaneous androgen metabolism: basic research and clinical perspectives. J Invest Dermatol, 2002. 119(5): p. 992-1007.
132. Baulieu, E.E., et al., Dehydroepiandrosterone (DHEA), DHEA sulfate, and aging: contribution of the DHEAge Study to a sociobiomedical issue. Proc Natl Acad Sci U S A, 2000. 97(8): p. 4279-84.
133. Yoshida, K., et al., Effect of dehydroepiandrosterone sulphate, oestrogens and prostaglandins on collagen metabolism in human cervical tissue in relation to cervical ripening. J Int Med Res, 1993. 21(1): p. 26-35.
134. Shin, M.H., et al., Modulation of collagen metabolism by the topical application of dehydroepiandrosterone to human skin. J Invest Dermatol, 2005. 124(2): p. 315-23.
135. Lee, K.S., K.Y. Oh, and B.C. Kim, Effects of dehydroepiandrosterone on collagen and collagenase gene expression by skin fibroblasts in culture. J Dermatol Sci, 2000. 23(2): p. 103-10.
136. S., F. and H.I. Maibach, Gender Differences in Skin, in Textbook of Aging Skin, M.A. Farage , K.W. Miller, and H.I. Maibach, Editors. 2010, Springer Berlin Heidelberg. p. 999-1017.
137. Calvo, E., et al., Pangenomic changes induced by DHEA in the skin of postmenopausal women. J Steroid Biochem Mol Biol, 2008. 112(4-5): p. 186-93.



